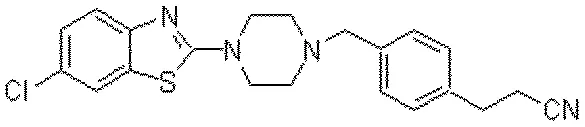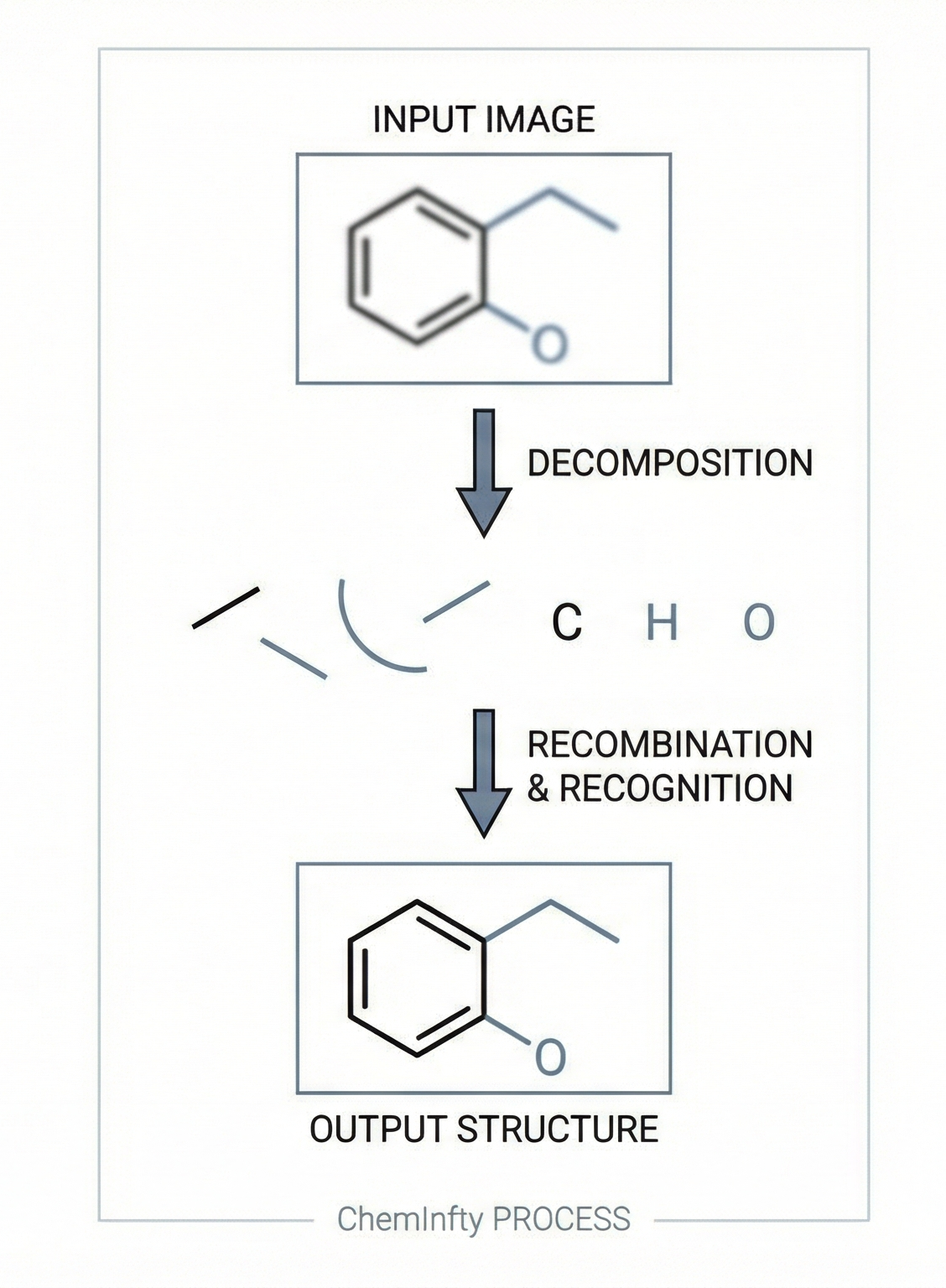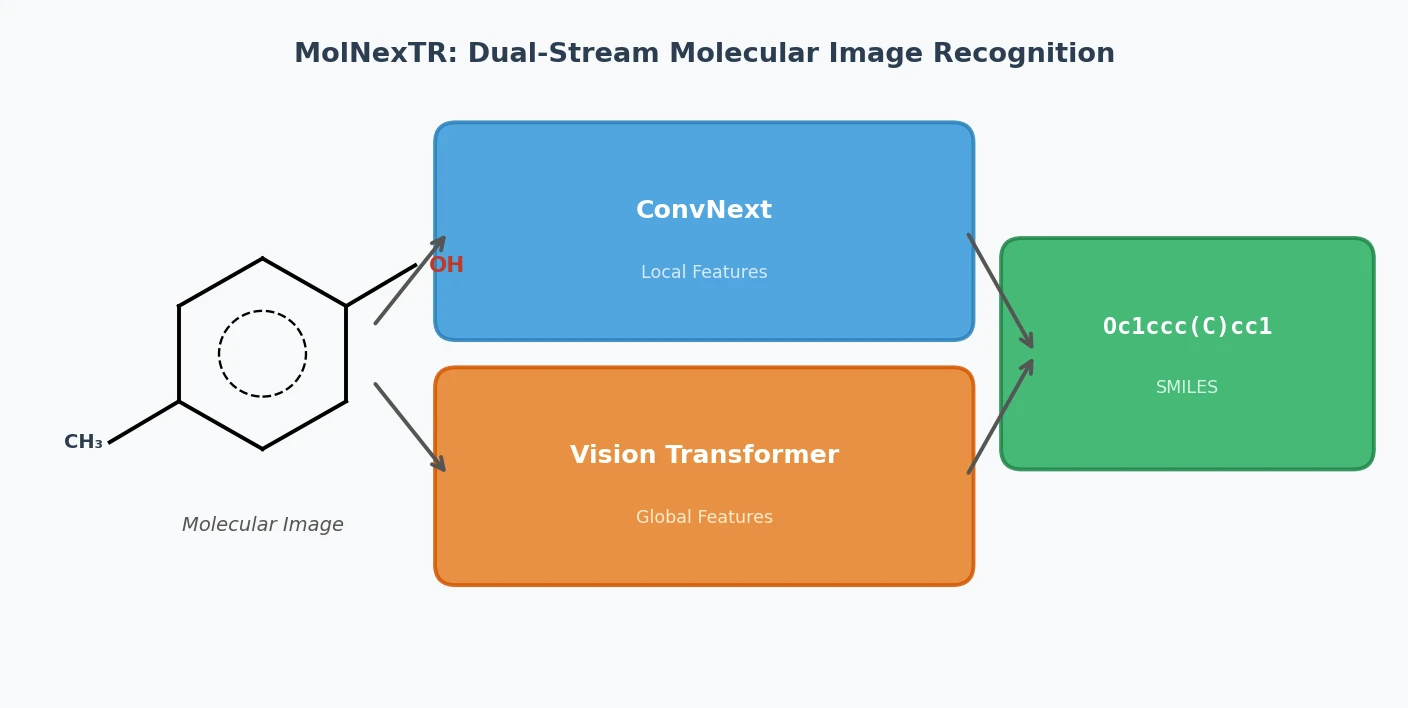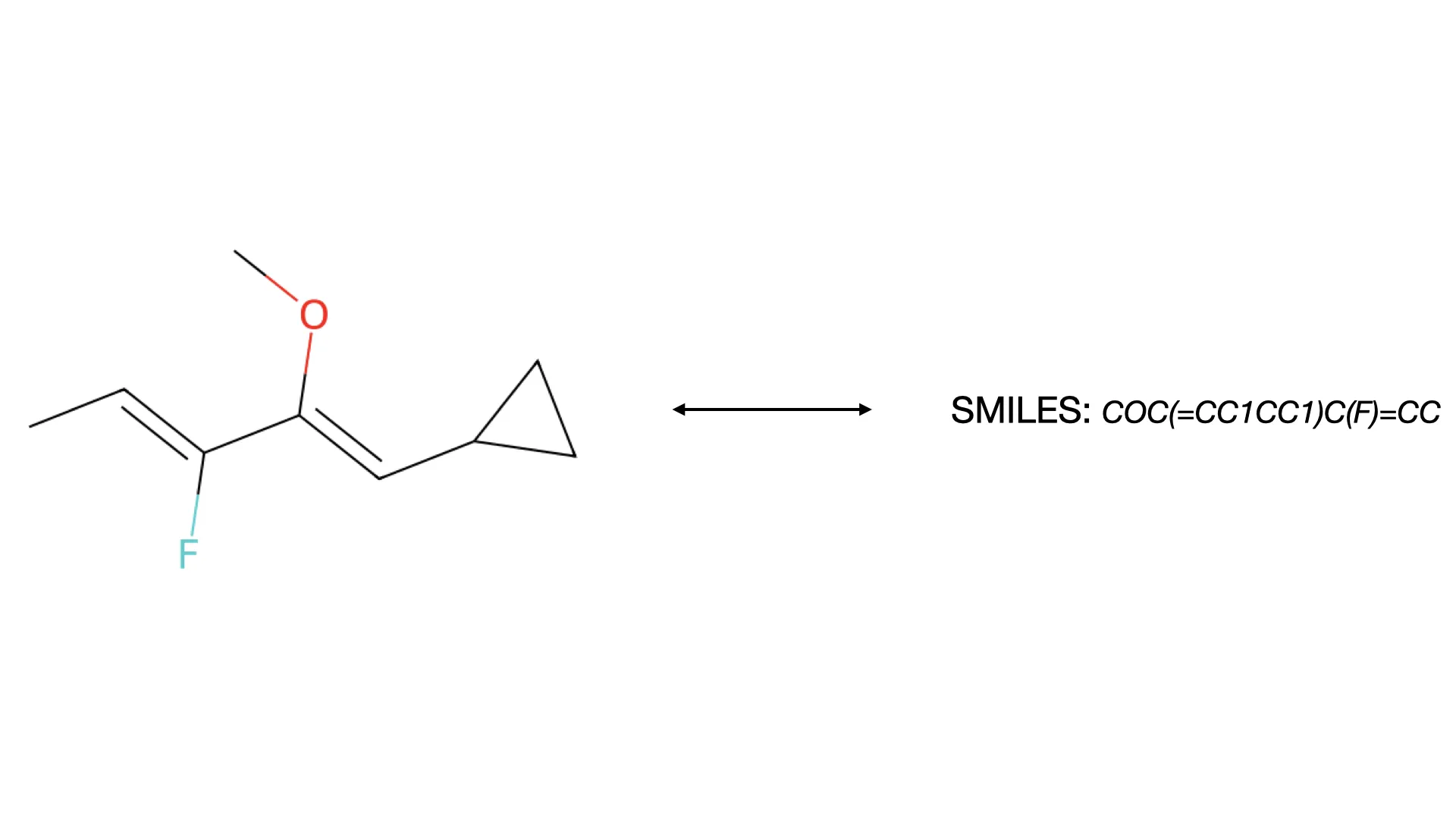
IMG2SMI: Translating Molecular Structure Images to SMILES
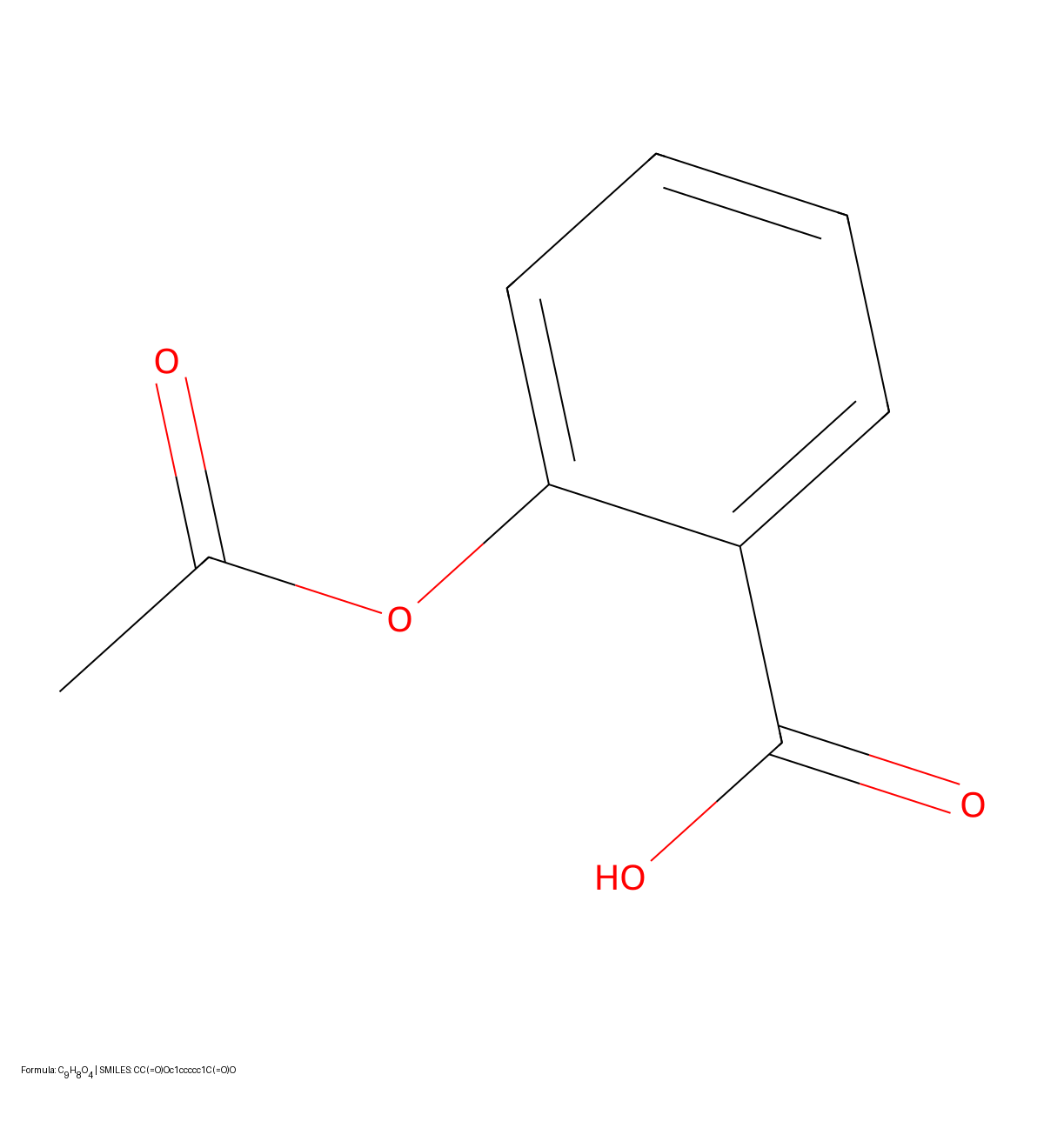
A 2021 image-to-text approach treating OCSR as an image captioning task. It uses Transformers with SELFIES representation to convert molecular structure diagrams into SMILES strings, enabling extraction of visual chemical knowledge from scientific literature.