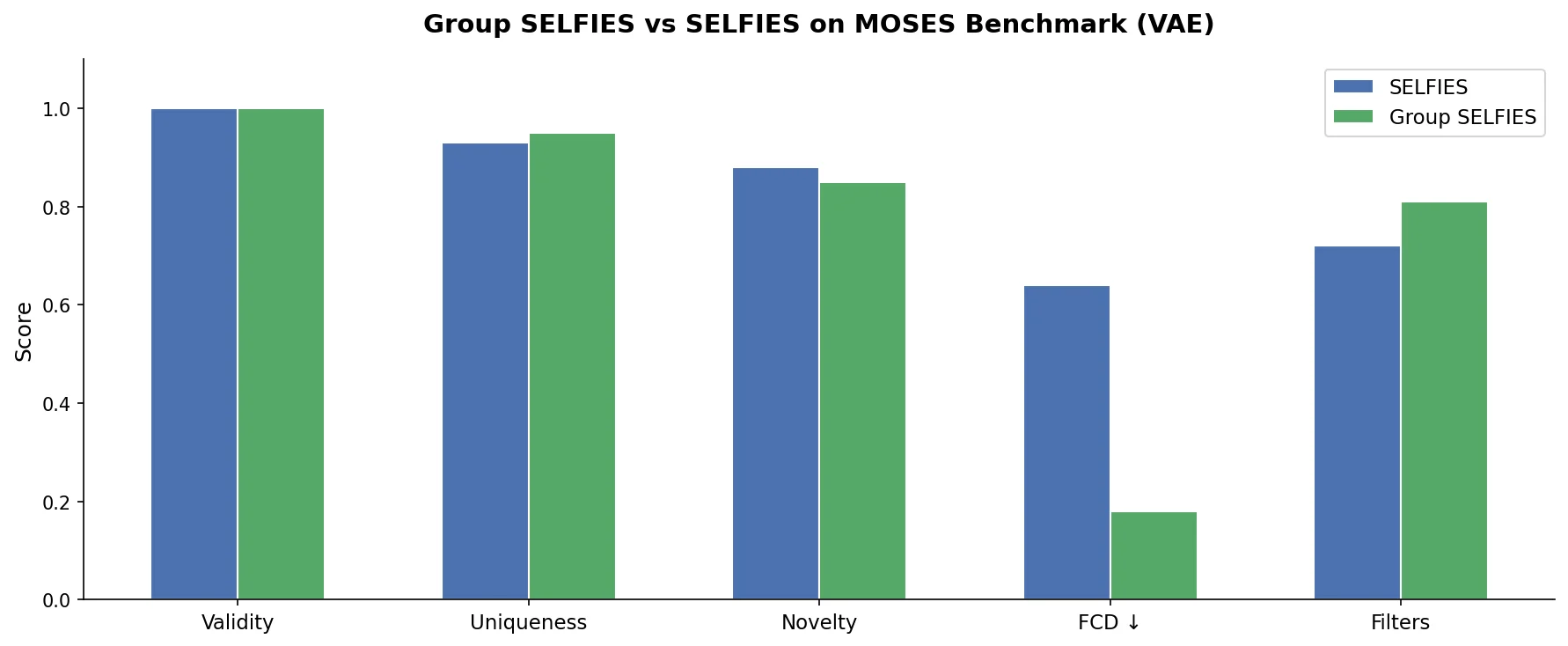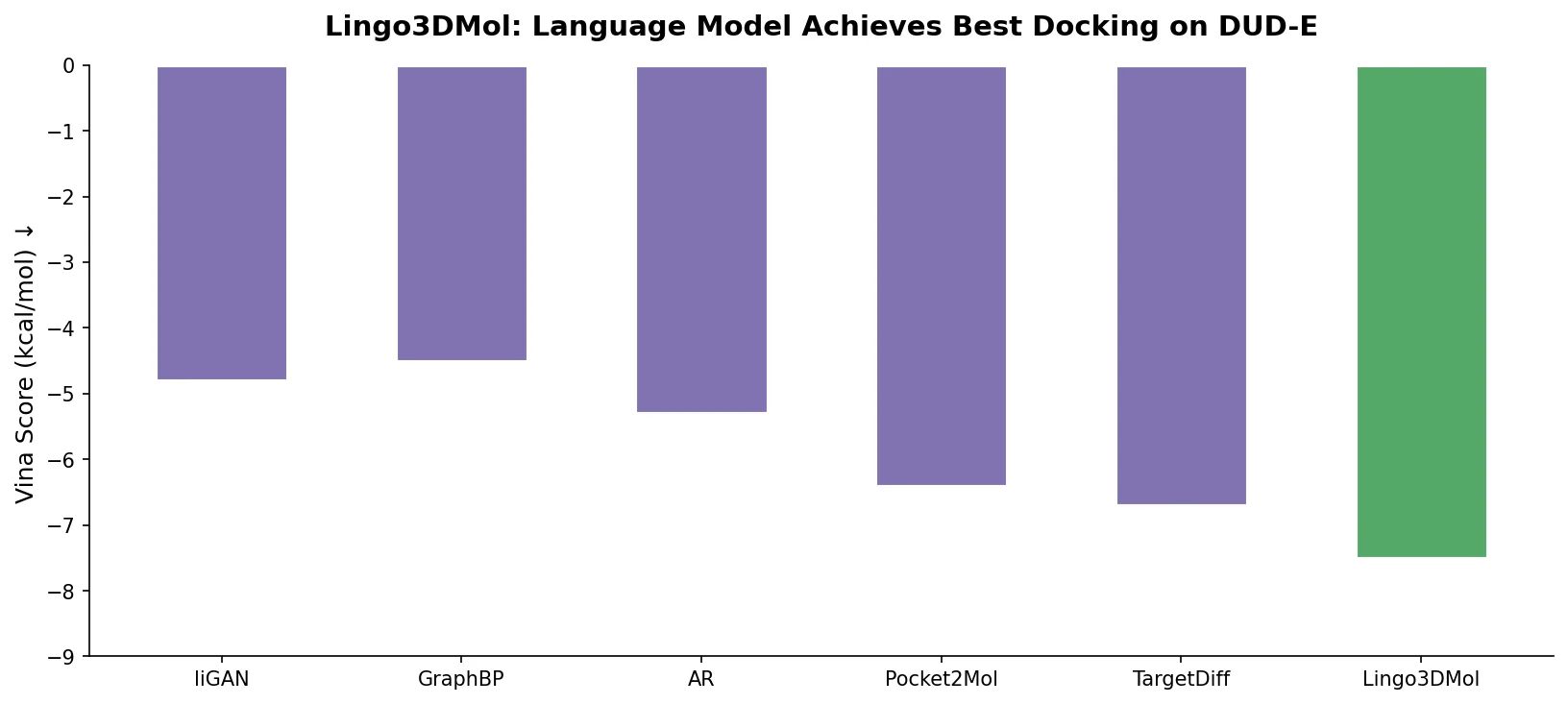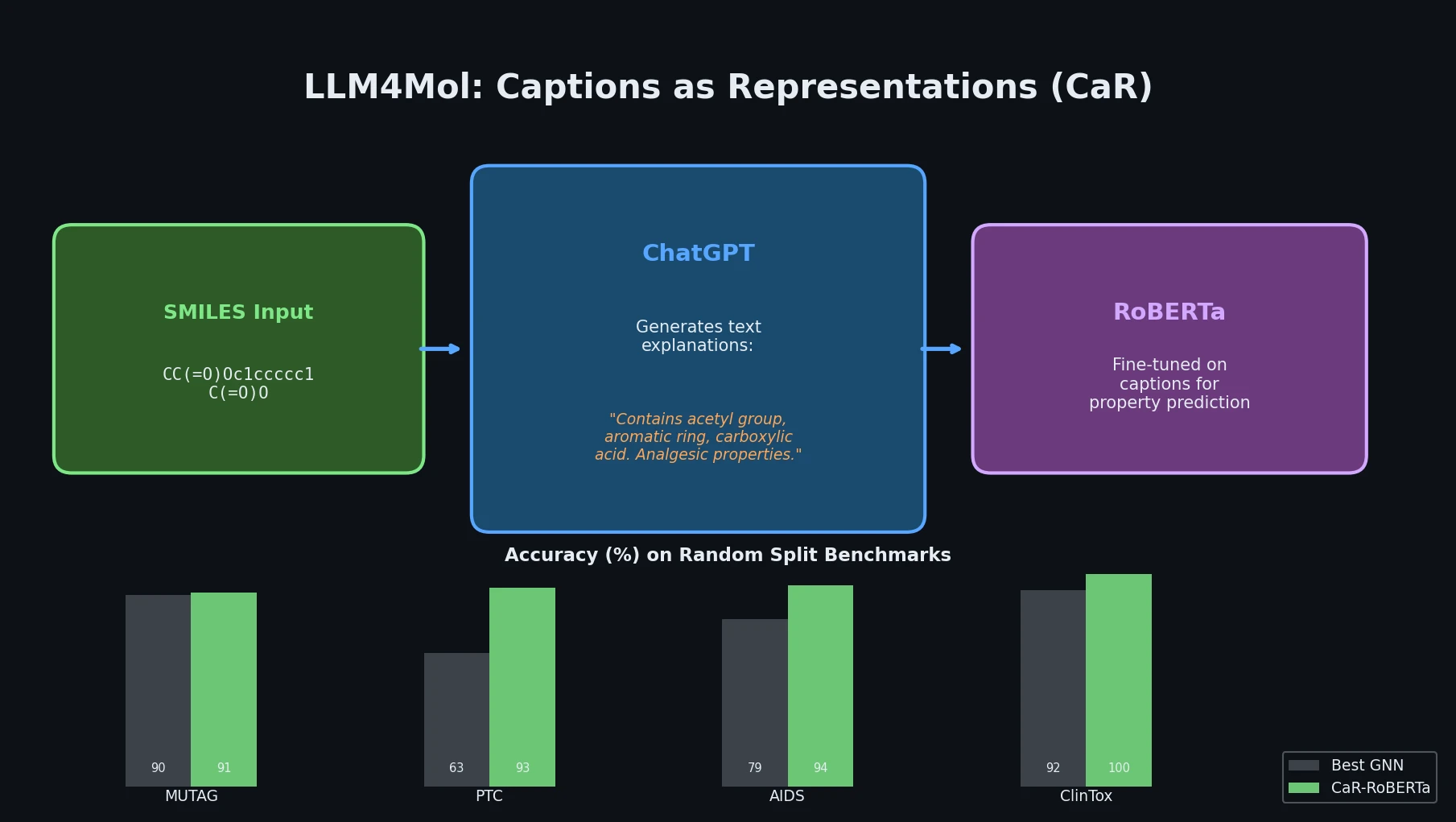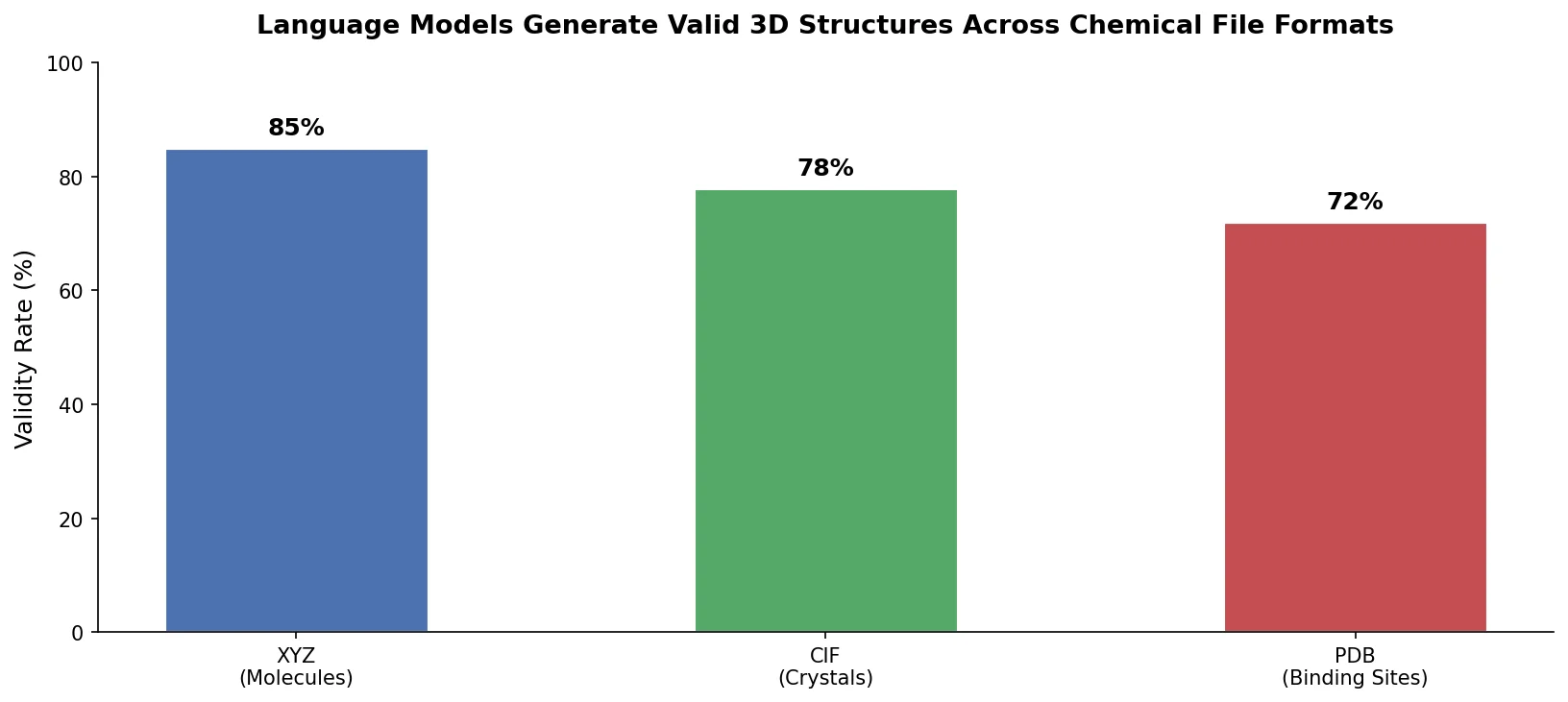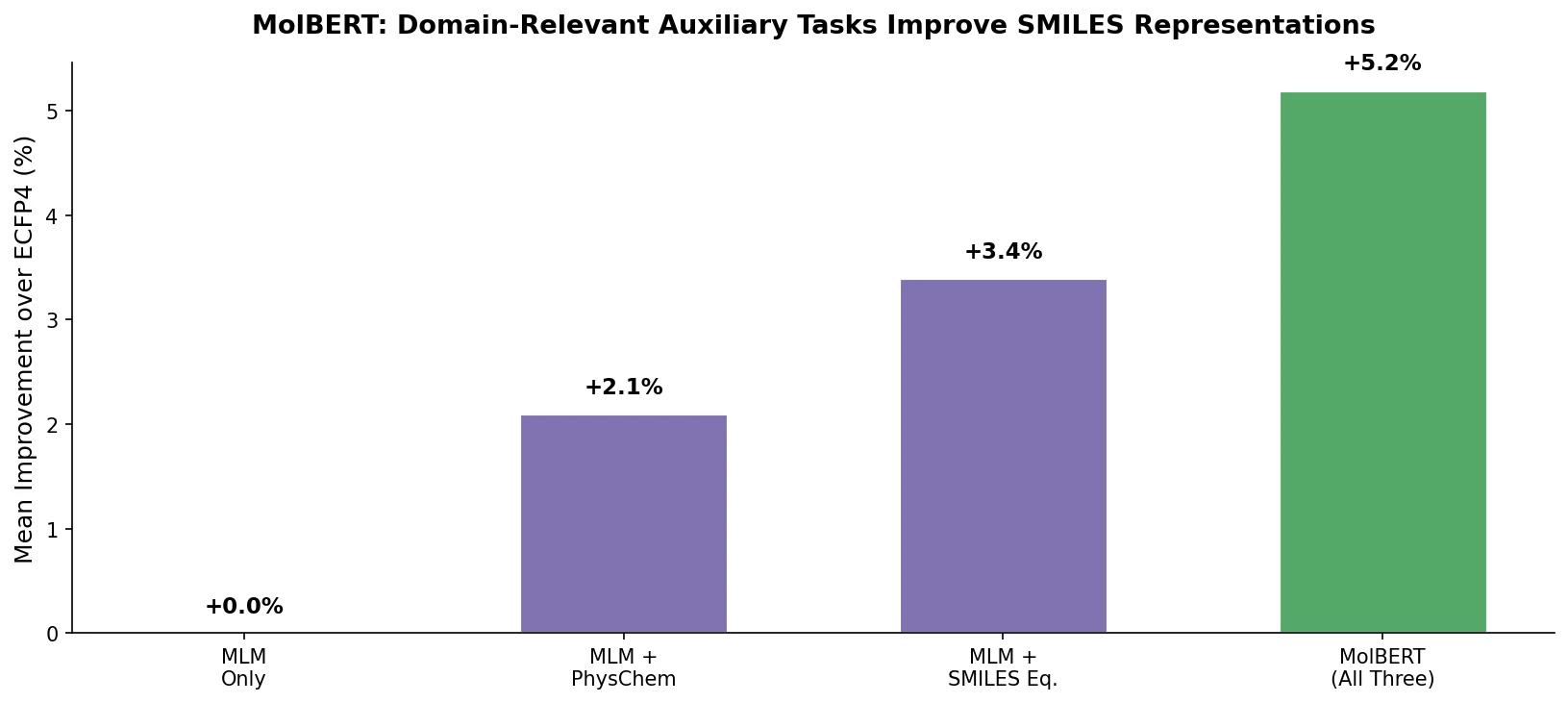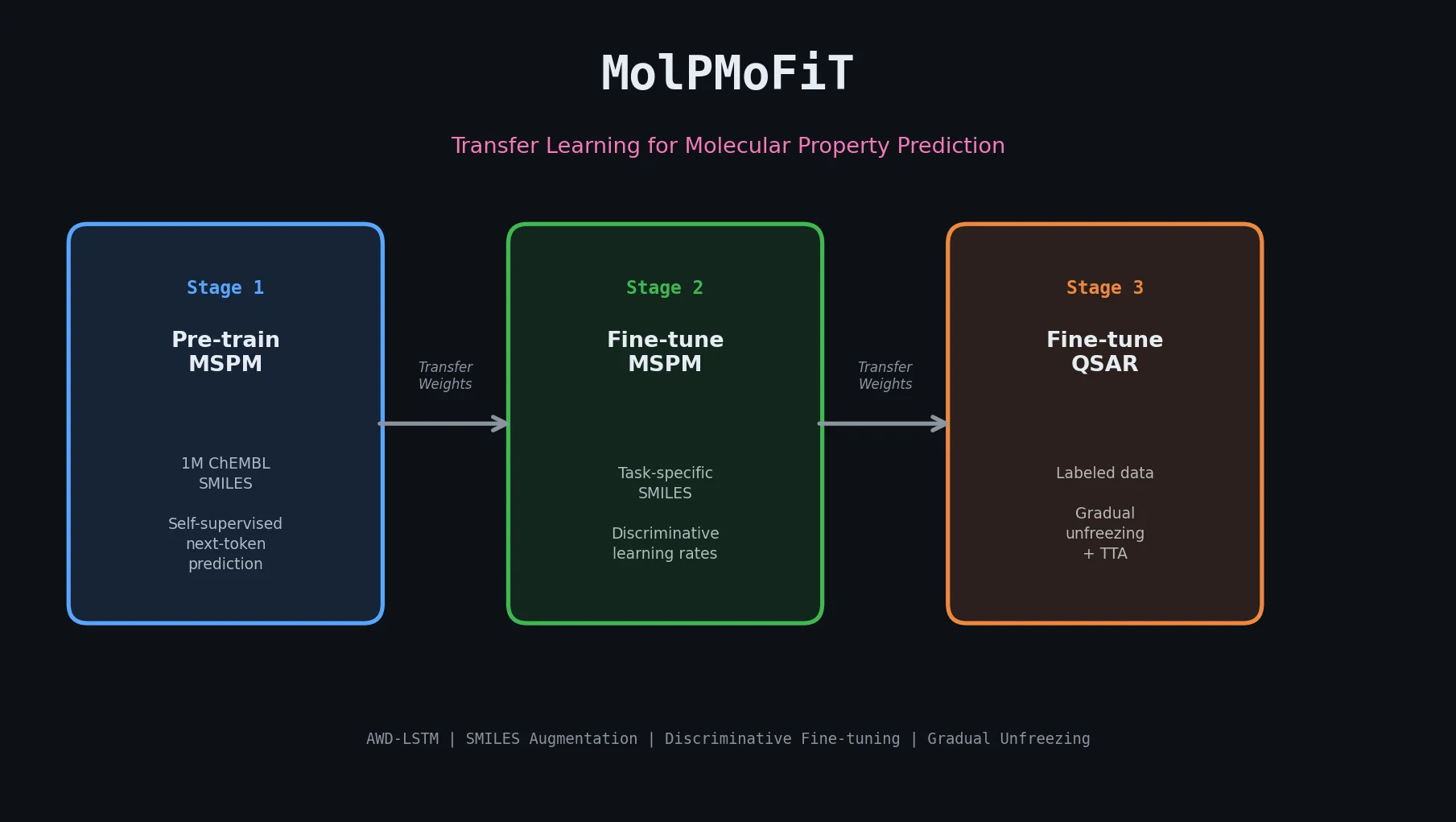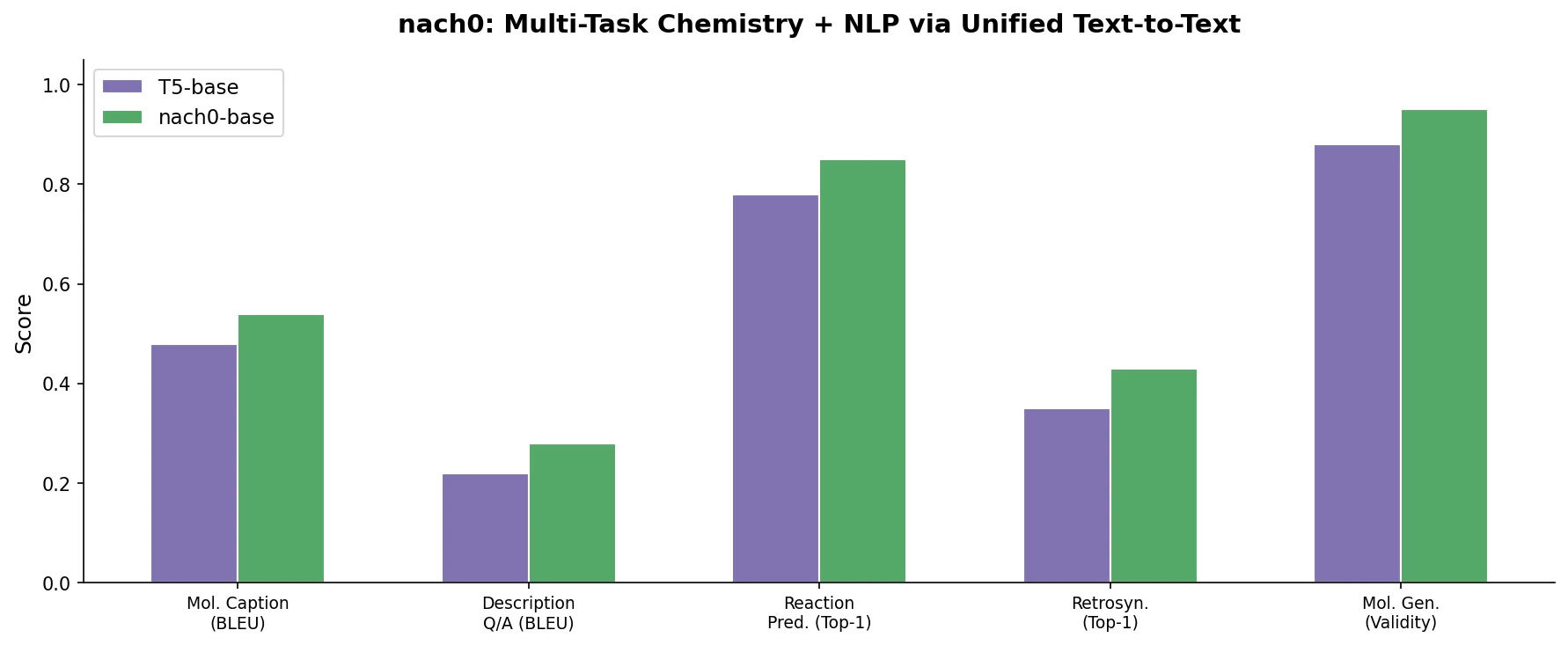
Fine-Tuning GPT-3 for Molecular Property Prediction
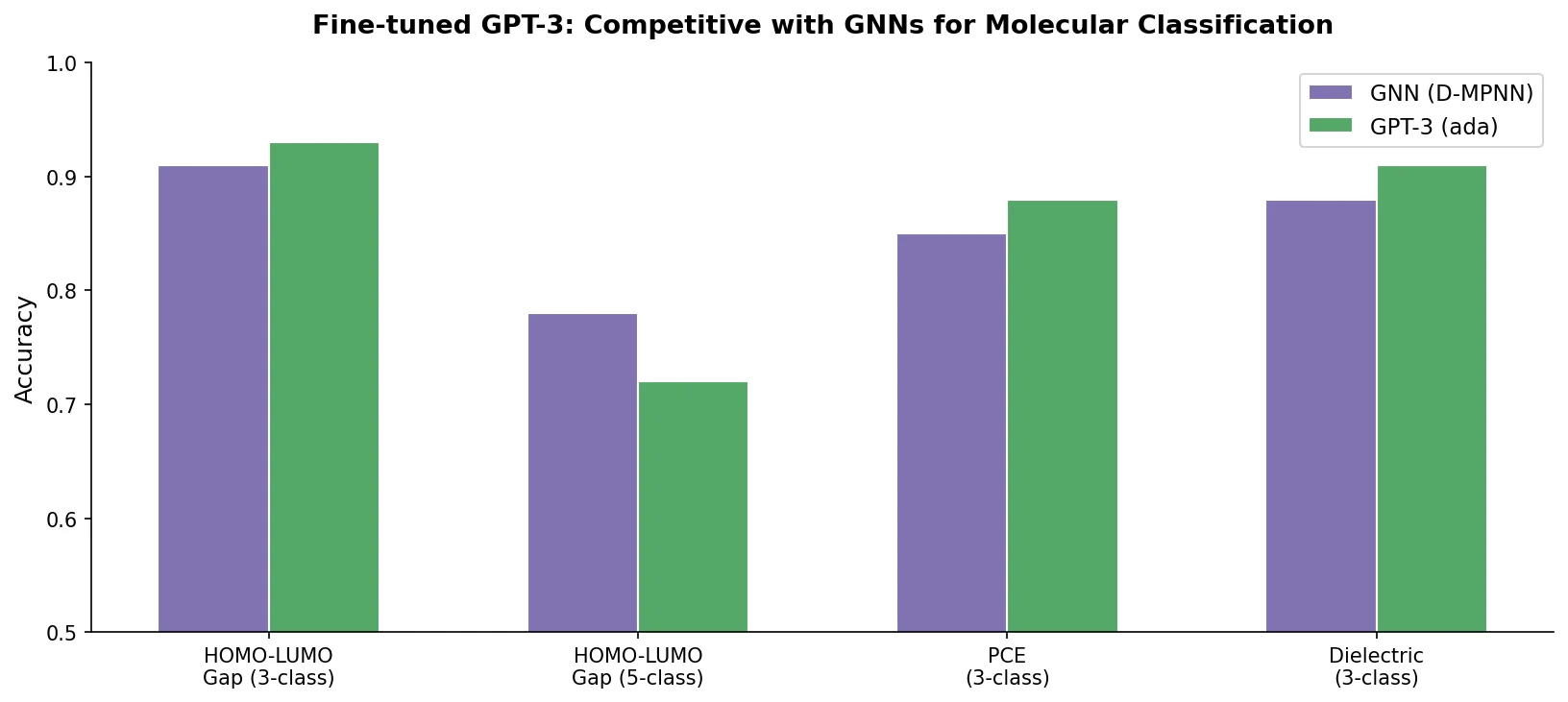
This paper fine-tunes GPT-3’s ada model on SMILES strings for classifying electronic properties (HOMO, LUMO) of organic semiconductor molecules, finding competitive accuracy with graph neural networks and exploring robustness through ablation studies.