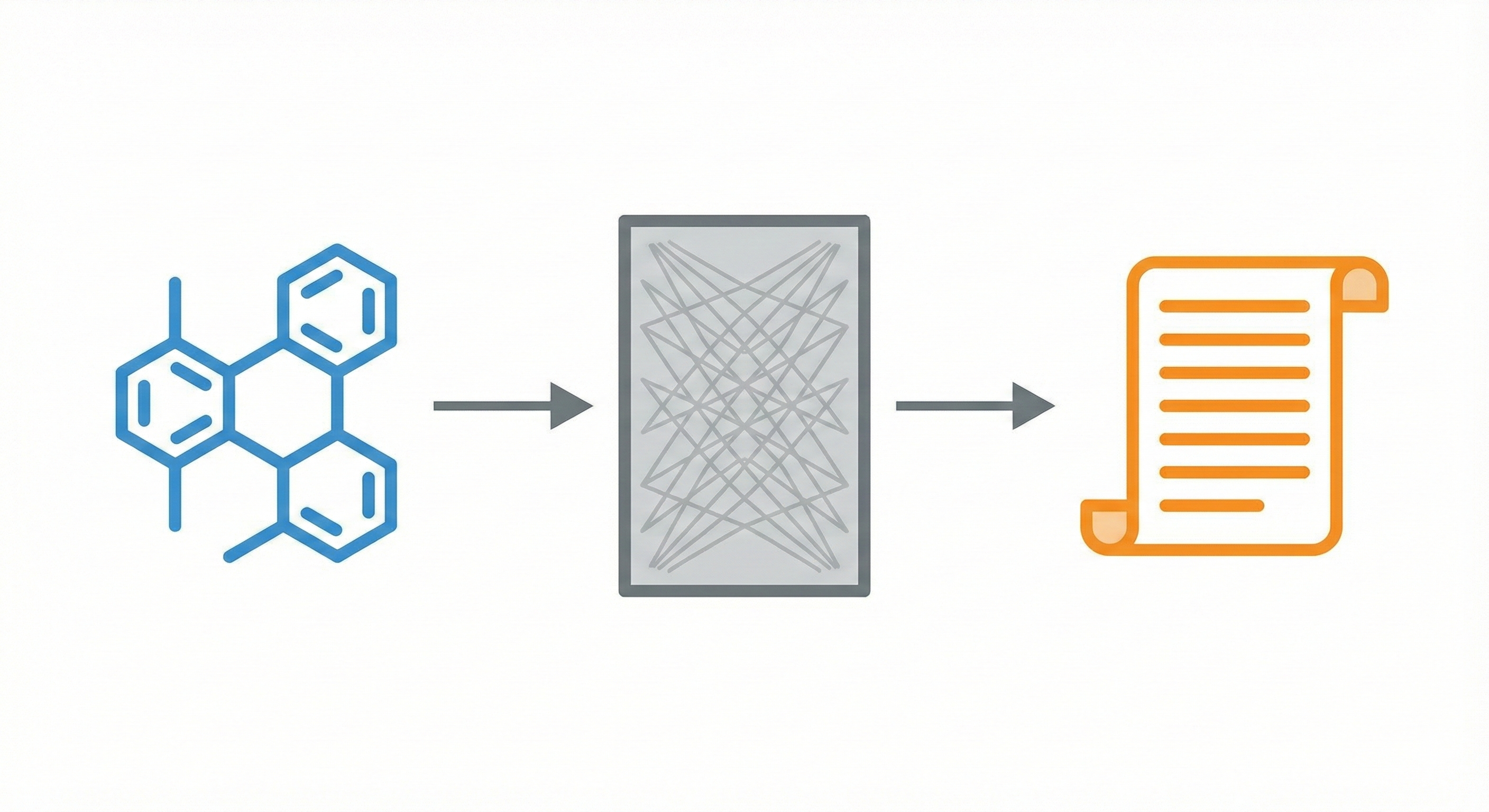
STOUT V2.0: Transformer-Based SMILES to IUPAC Translation
STOUT V2.0 uses Transformers trained on ~1 billion SMILES-IUPAC pairs to accurately translate chemical structures into systematic names (and vice-versa), outperforming its RNN predecessor.

STOUT V2.0 uses Transformers trained on ~1 billion SMILES-IUPAC pairs to accurately translate chemical structures into systematic names (and vice-versa), outperforming its RNN predecessor.
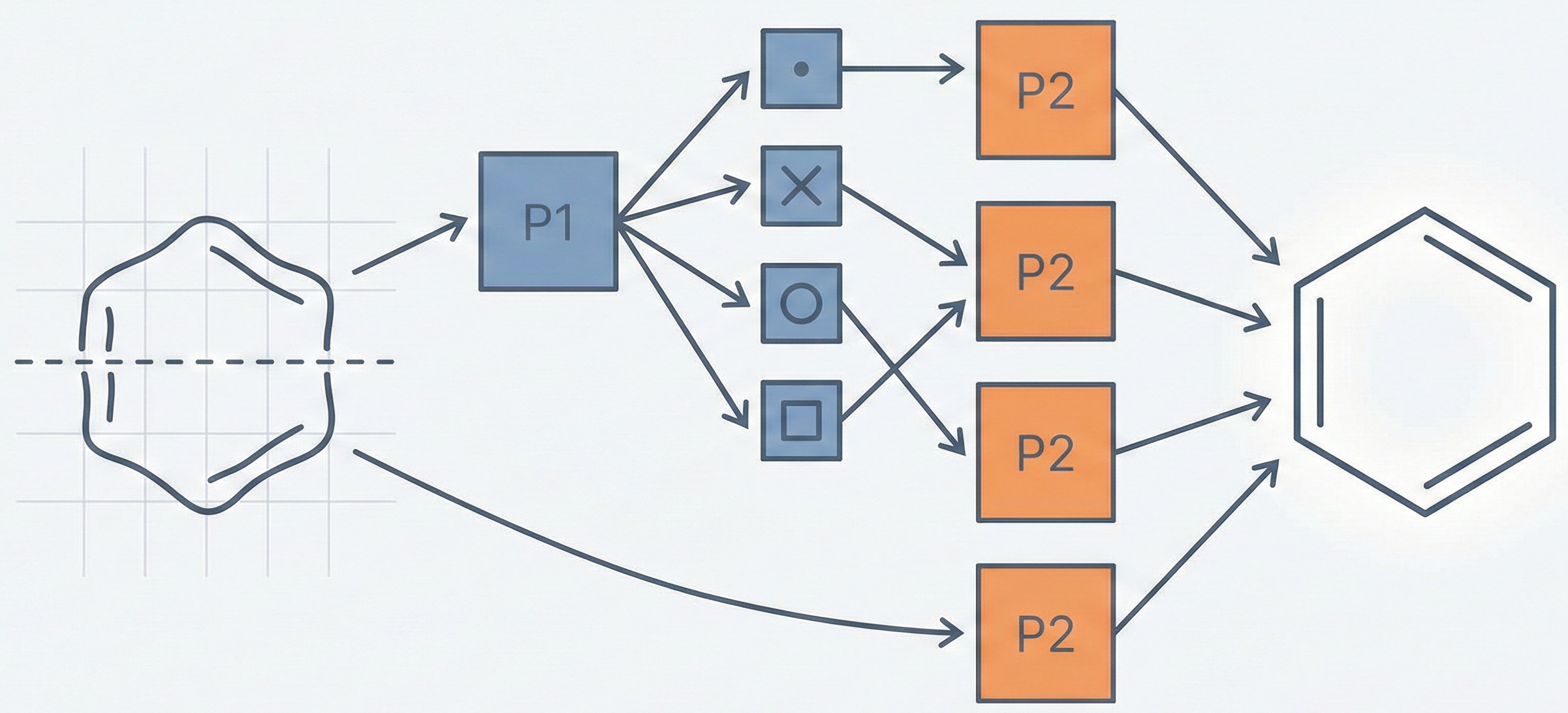
Proposes a specialized Classifier-Recognizer architecture that first categorizes rings by heteroatom (S, N, O) and then identifies the specific ring using optimized grid inputs.
A 2013 paper introducing a hybrid recognition system for handwritten chemical symbols on touch devices. Combines Support Vector Machines (SVM) for classification with elastic matching for geometric verification, achieving 89.7% top-1 accuracy on pen-based input for chemical structure drawing applications.
HMM-based method for recognizing online handwritten chemical symbols using 11-dimensional local features including derivatives, curvature, and linearity. Achieves 89.5% top-1 accuracy and 98.7% top-3 accuracy on a custom dataset of 64 chemical symbols.
Yang et al. propose a two-level recognition system for handwritten chemical formulas, combining global structural analysis to identify substances with local character recognition using ANNs, achieving ~96% accuracy on a dataset of 1197 expressions.
A three-level grammatical framework (formula, molecule, text) for parsing online handwritten chemical formulas, generating semantic graphs that capture both connectivity and layout using context-free grammars and HMMs.
Proposes a novel two-level algorithm for on-line handwritten chemical expression recognition, combining substance-level matching with character-level segmentation to achieve 96% accuracy.
This paper proposes a double-stage architecture using SVM for rough classification and HMM for fine recognition. It features a novel Point Sequence Reordering (PSR) algorithm that significantly improves accuracy on organic ring structures.
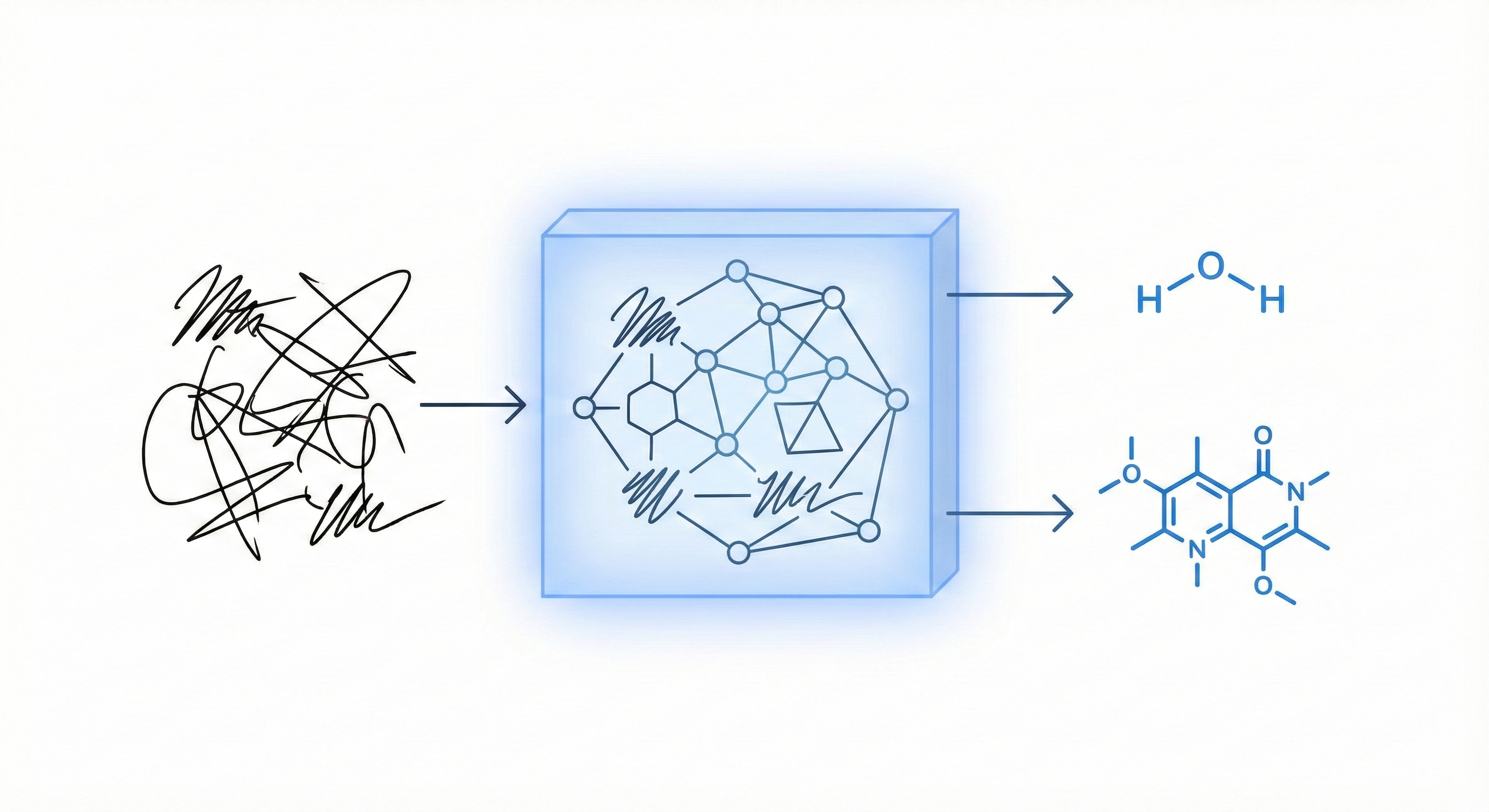
Proposes a unified statistical framework for recognizing both inorganic and organic handwritten chemical expressions. Introduces the Chemical Expression Structure Graph (CESG) and uses a weighted direction graph search for structural analysis, achieving 83.1% top-5 accuracy on a large proprietary dataset.
This paper introduces MLOCSR, a system that pipelines low-level image vectorization with a high-level probabilistic Markov Logic Network to recognize chemical structures. It replaces brittle heuristics with weighted logic rules, significantly outperforming state-of-the-art systems like OSRA on degraded or low-resolution images.
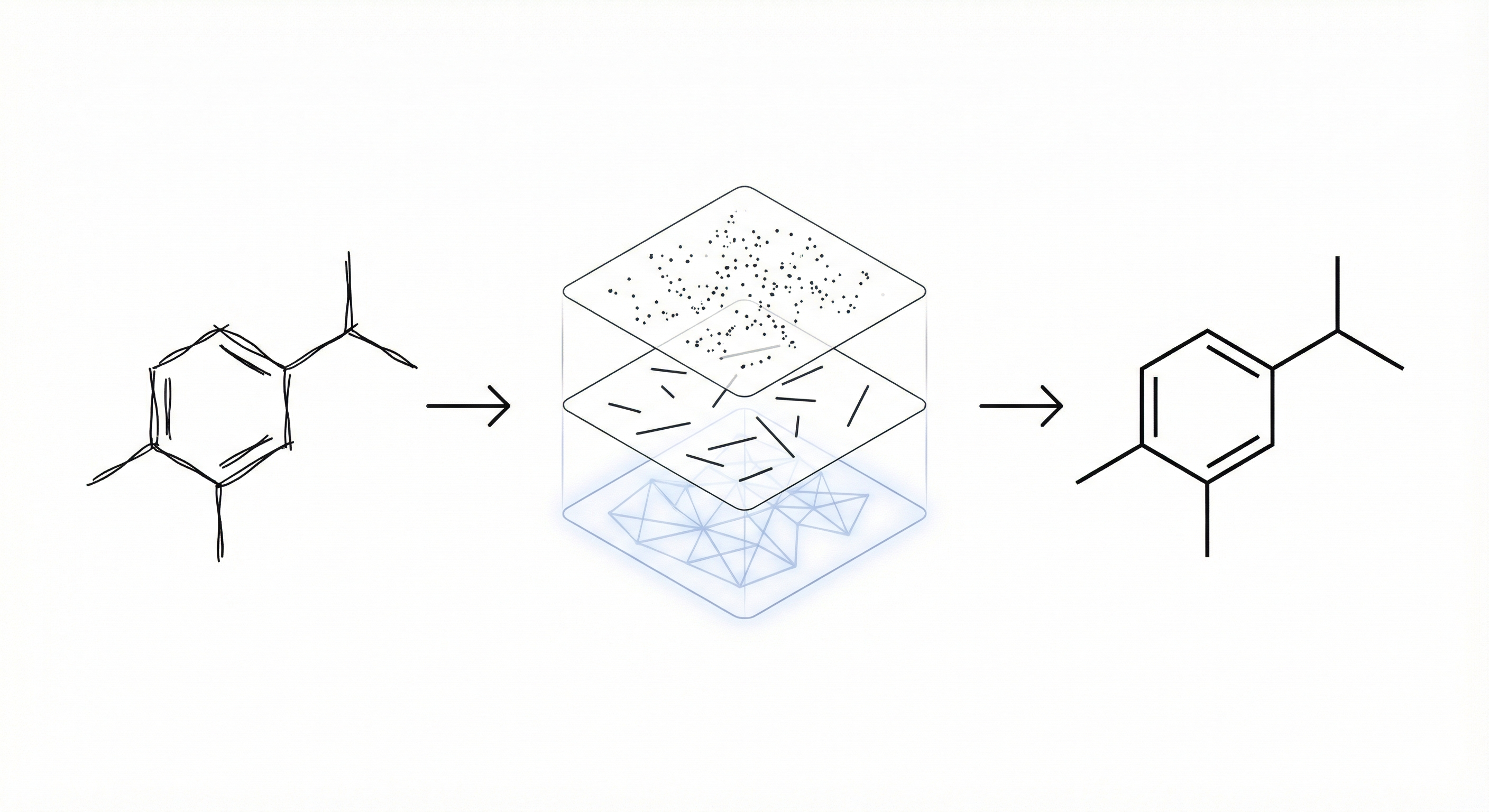
ChemInk introduces a sketch recognition system for chemical diagrams that combines multi-level visual features via a joint Conditional Random Field (CRF), achieving 97.4% accuracy and outperforming CAD tools in user speed.

Geoffrey Hinton’s 1984 technical report that formally derives the efficiency of distributed representations (coarse coding) and demonstrates their properties of automatic generalization, content-addressability, and robustness to damage.