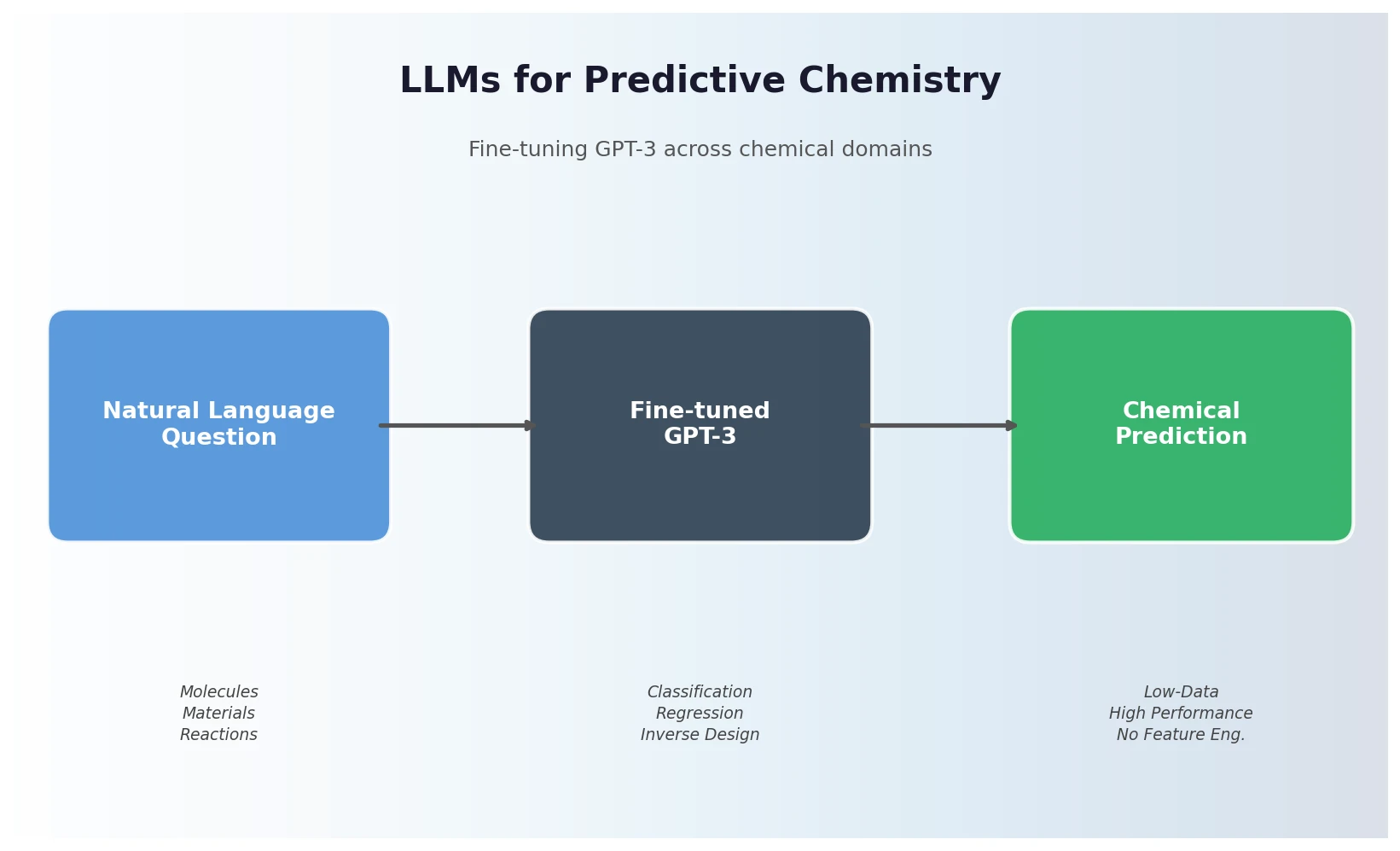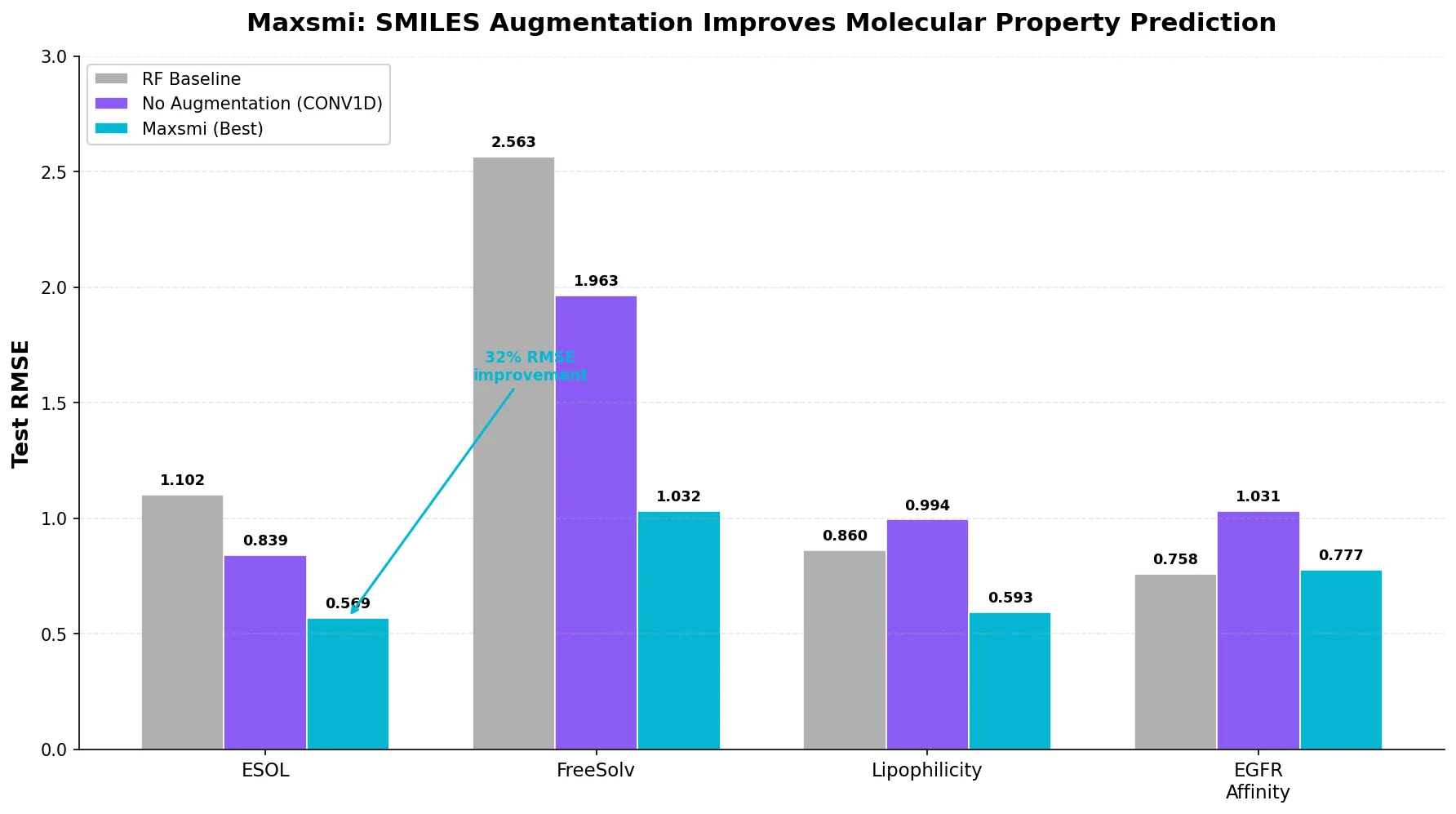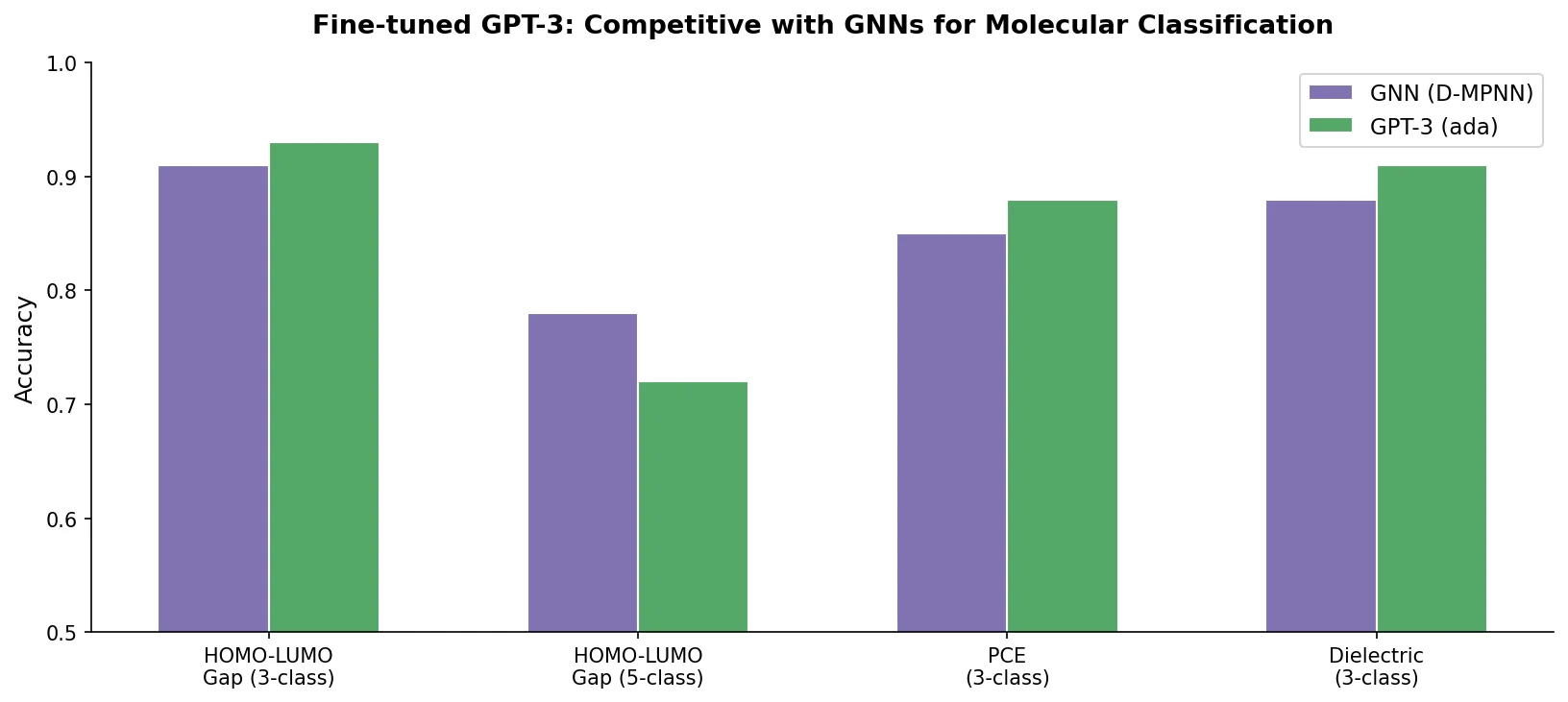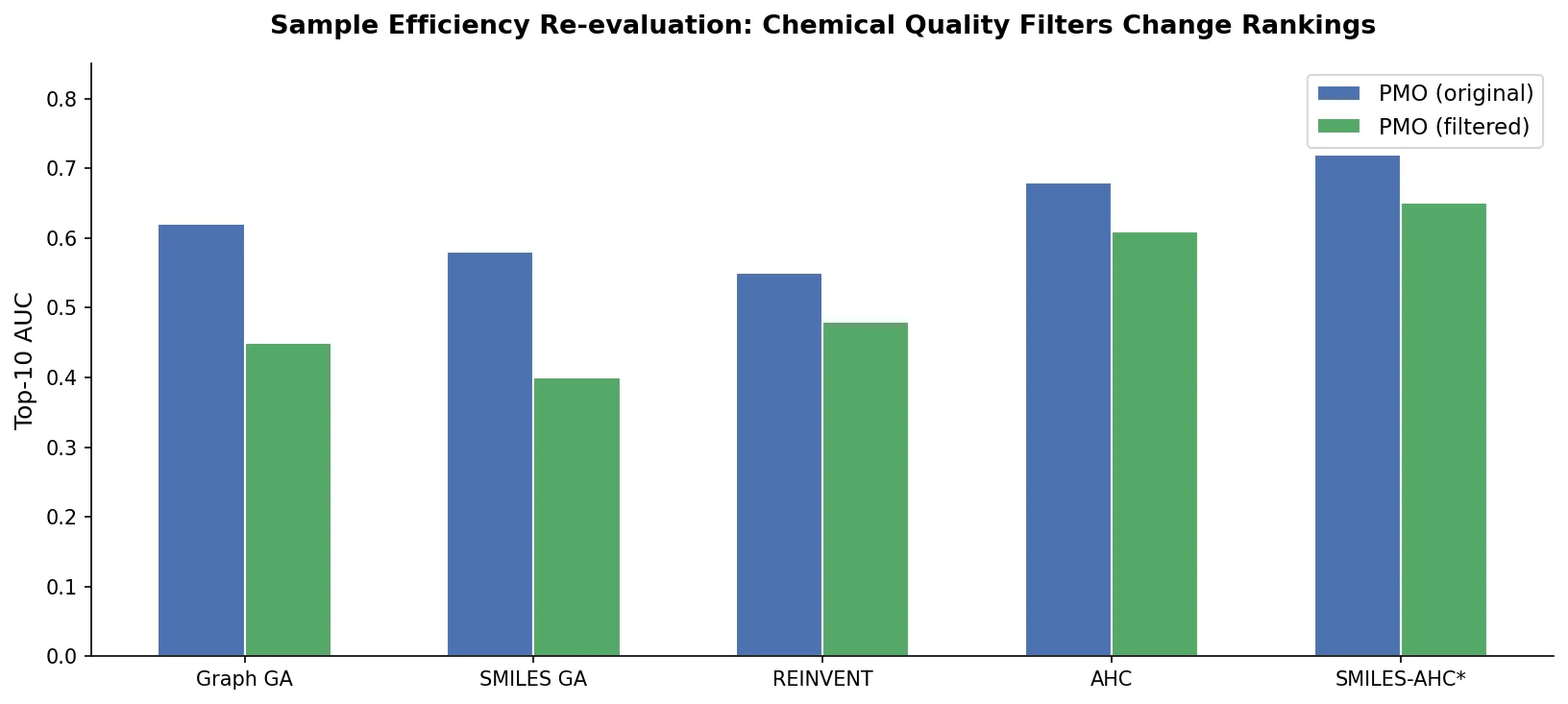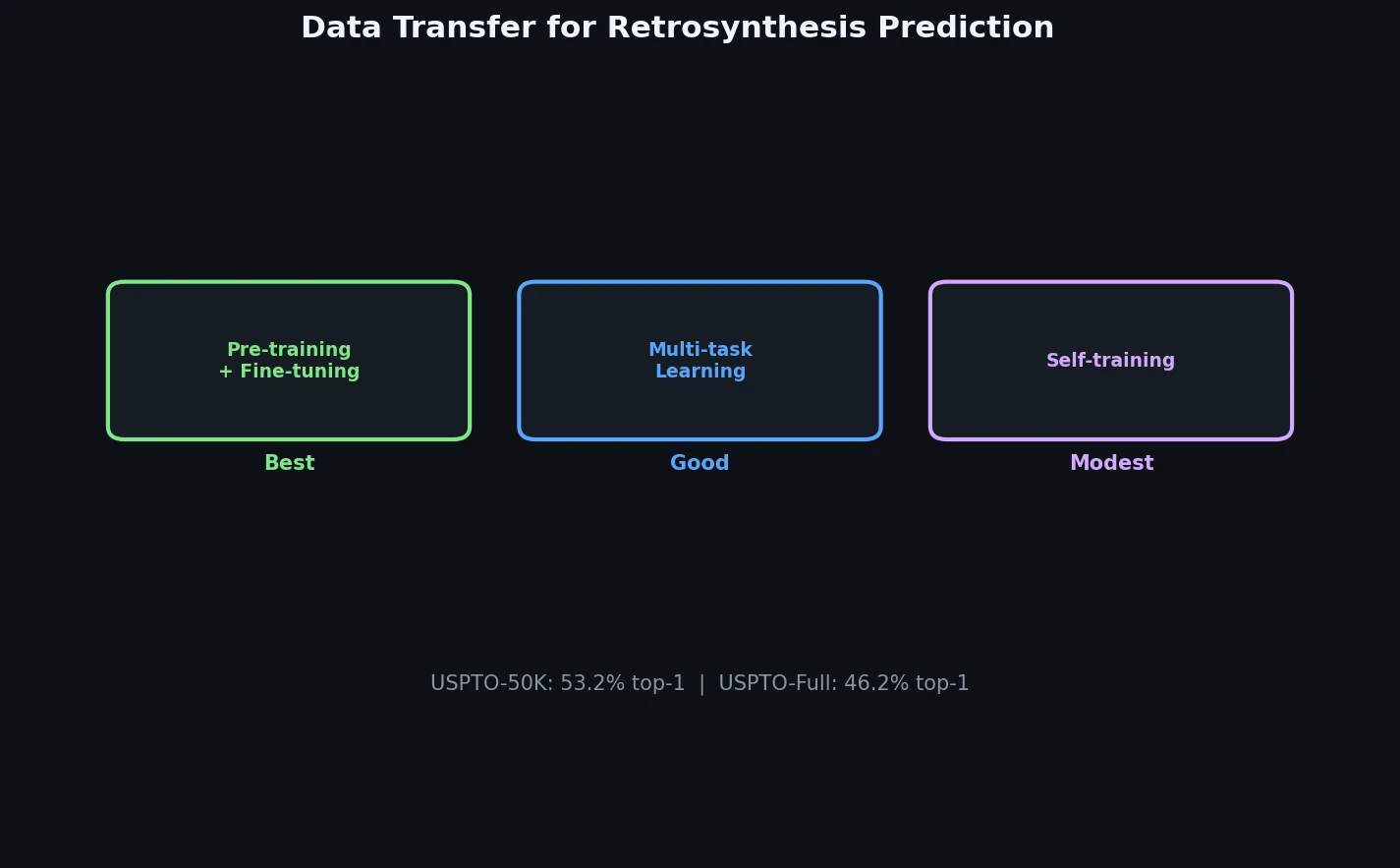
Data Transfer Approaches for Seq-to-Seq Retrosynthesis
A systematic study of data transfer techniques (joint training, self-training, pre-training plus fine-tuning) applied to Transformer-based retrosynthesis. Pre-training on USPTO-Full followed by fine-tuning on USPTO-50K achieves the best results, improving top-1 accuracy from 35.3% to 57.4%.