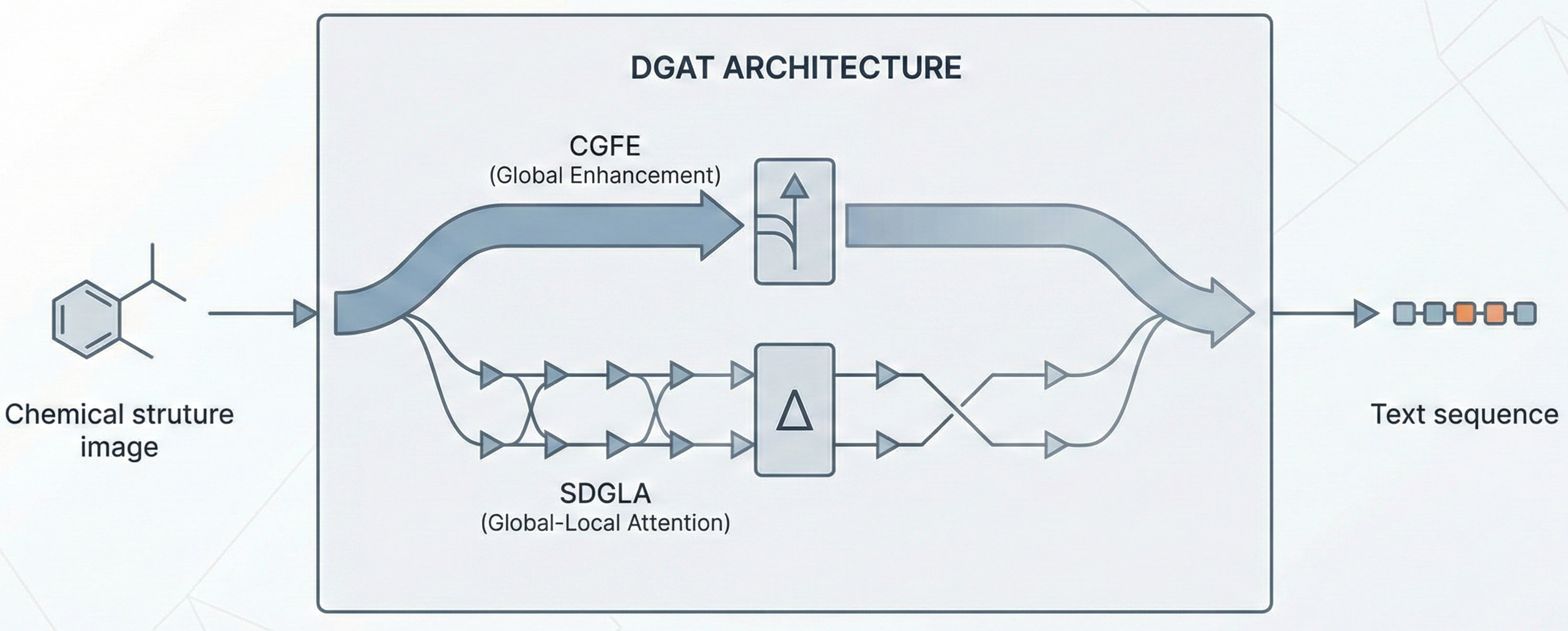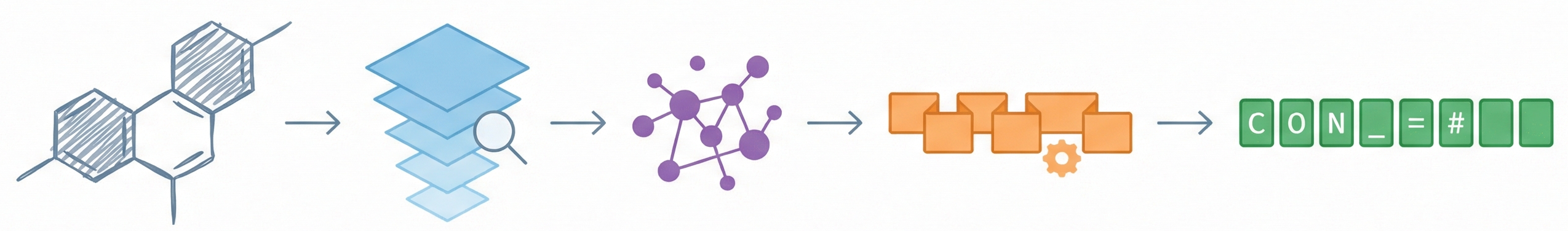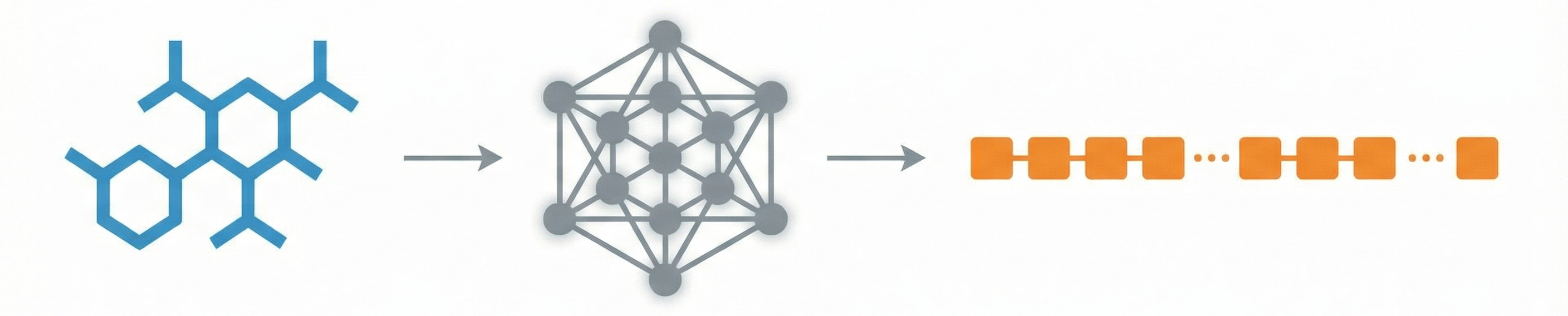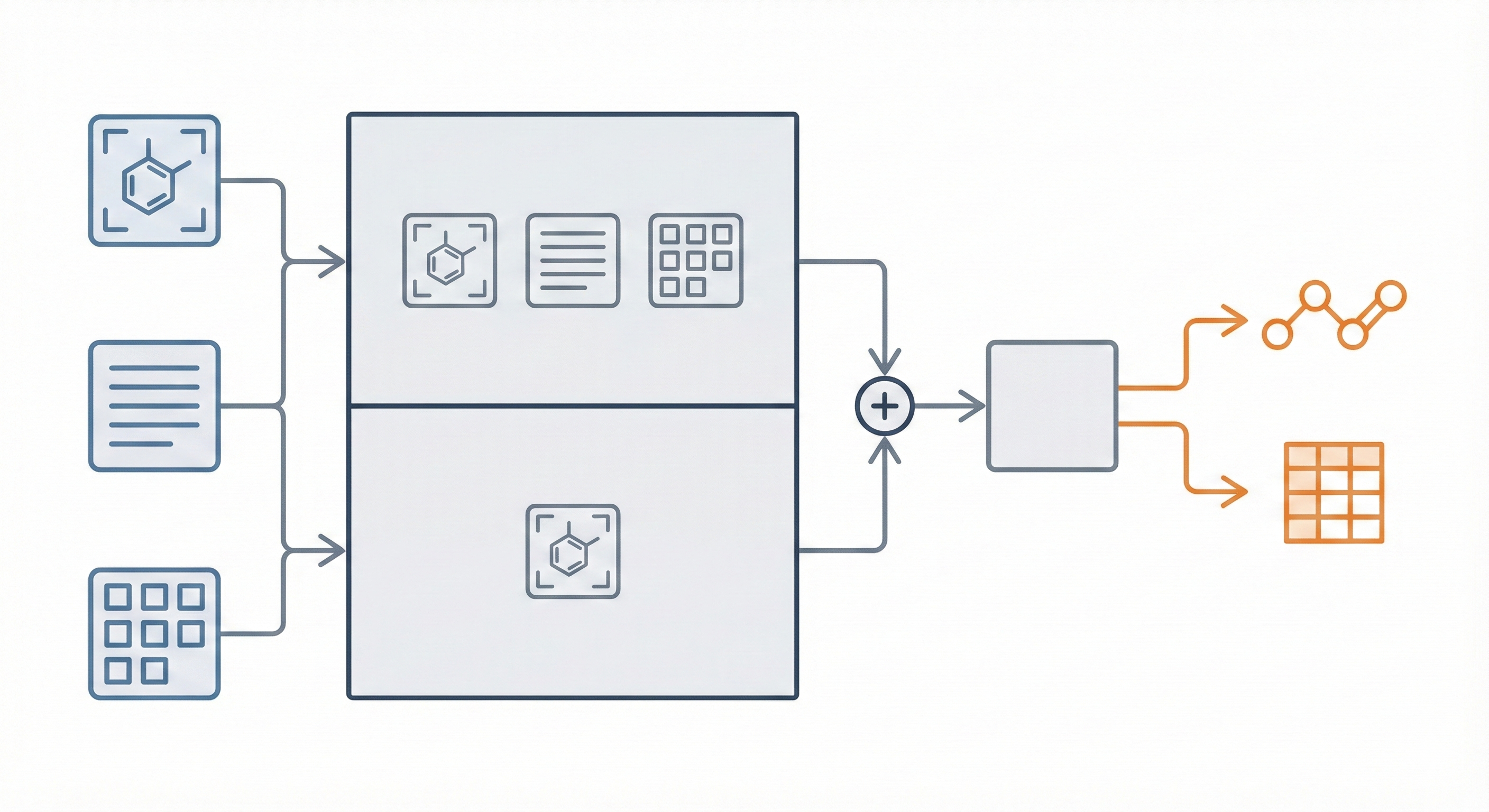
DECIMER.ai: Optical Chemical Structure Recognition
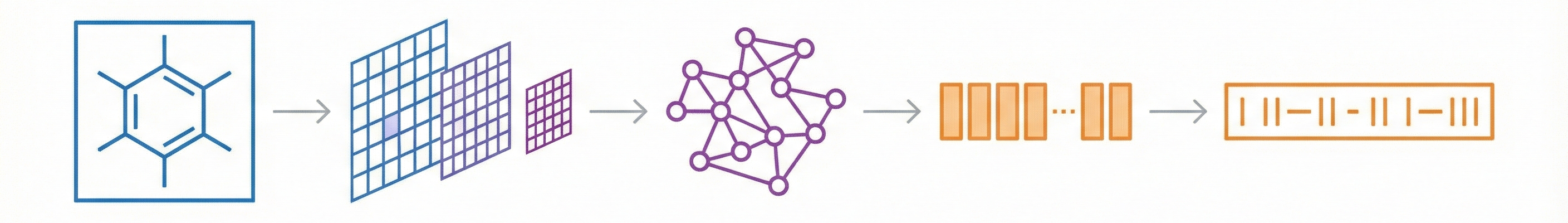
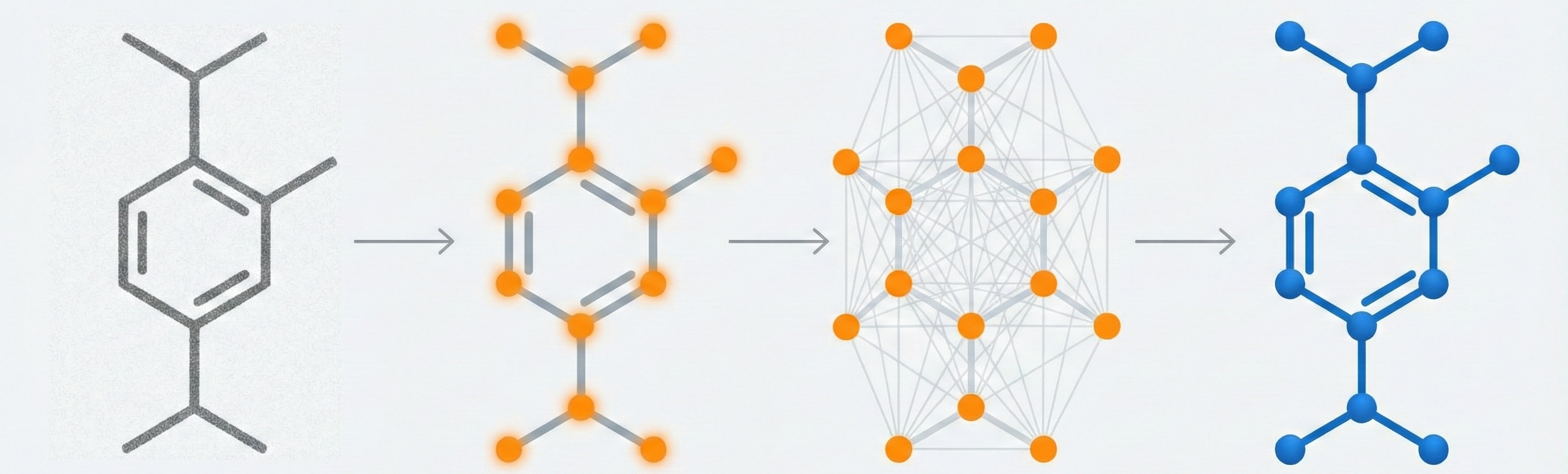
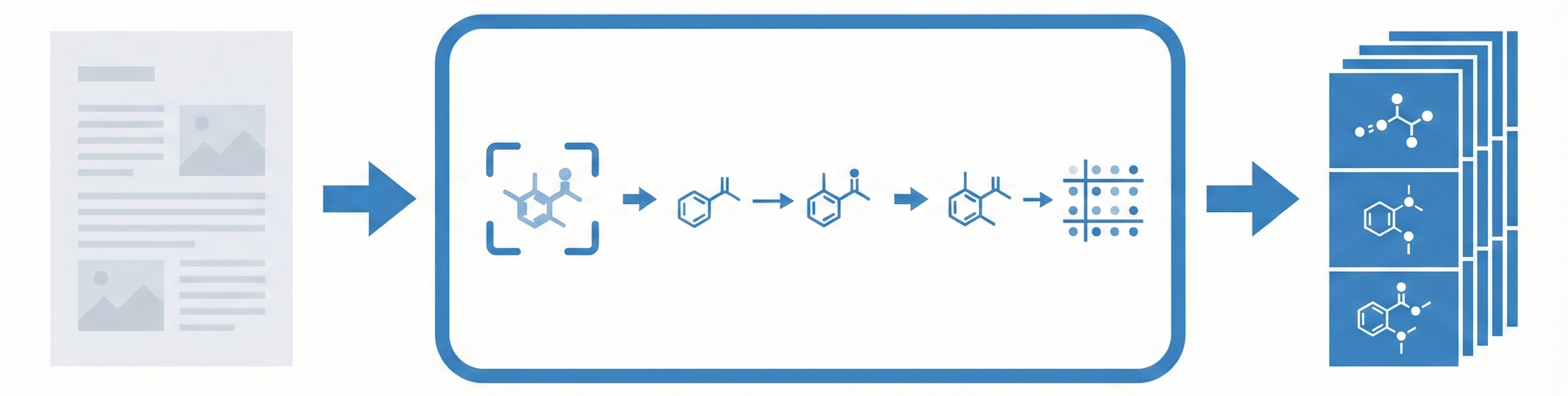
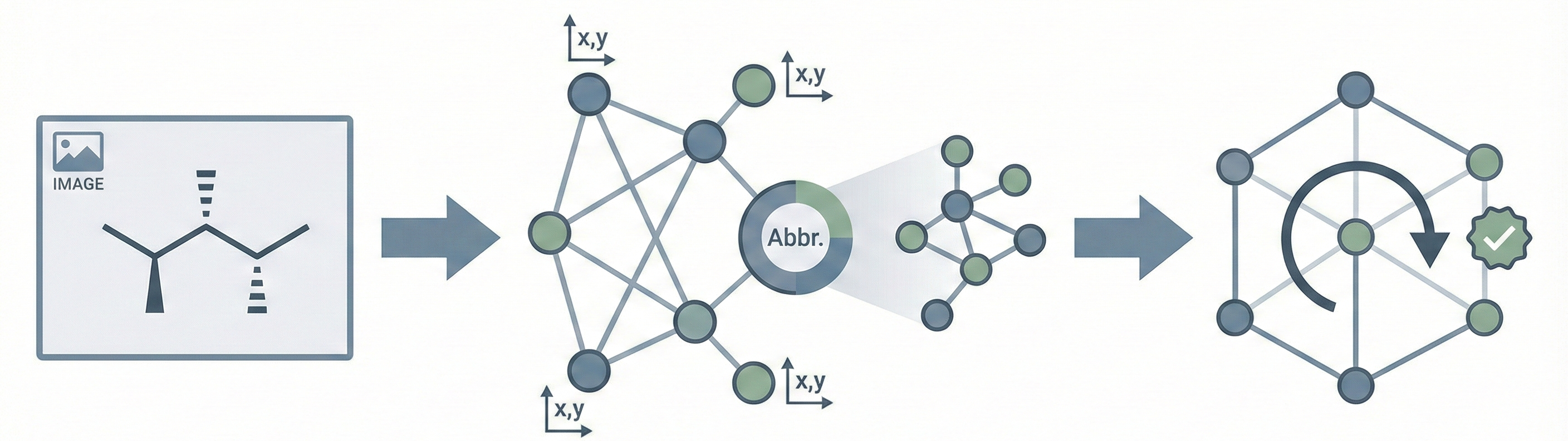
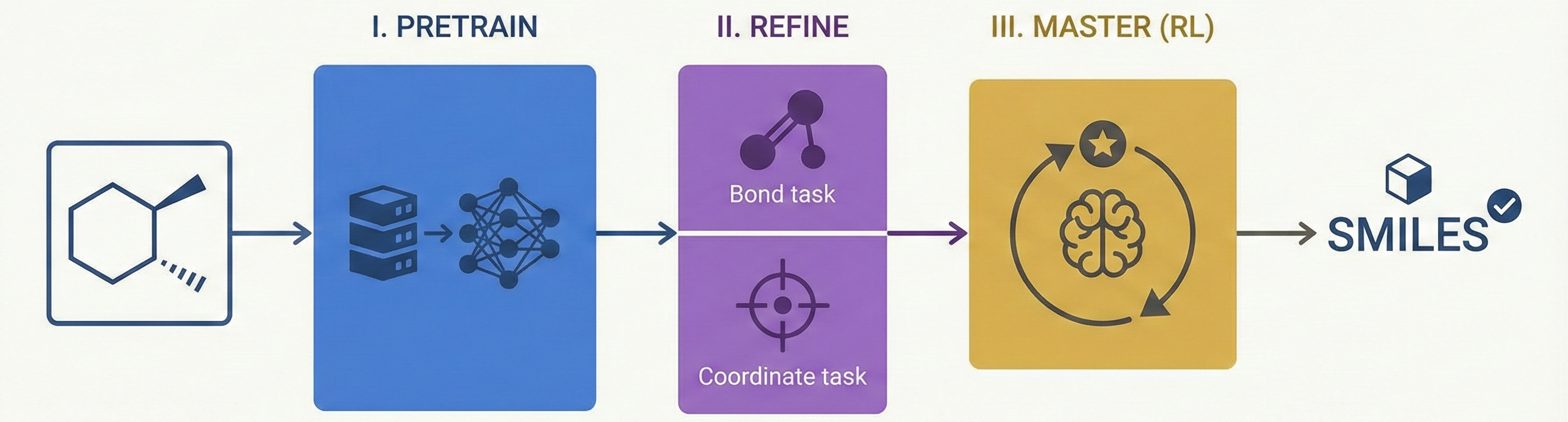
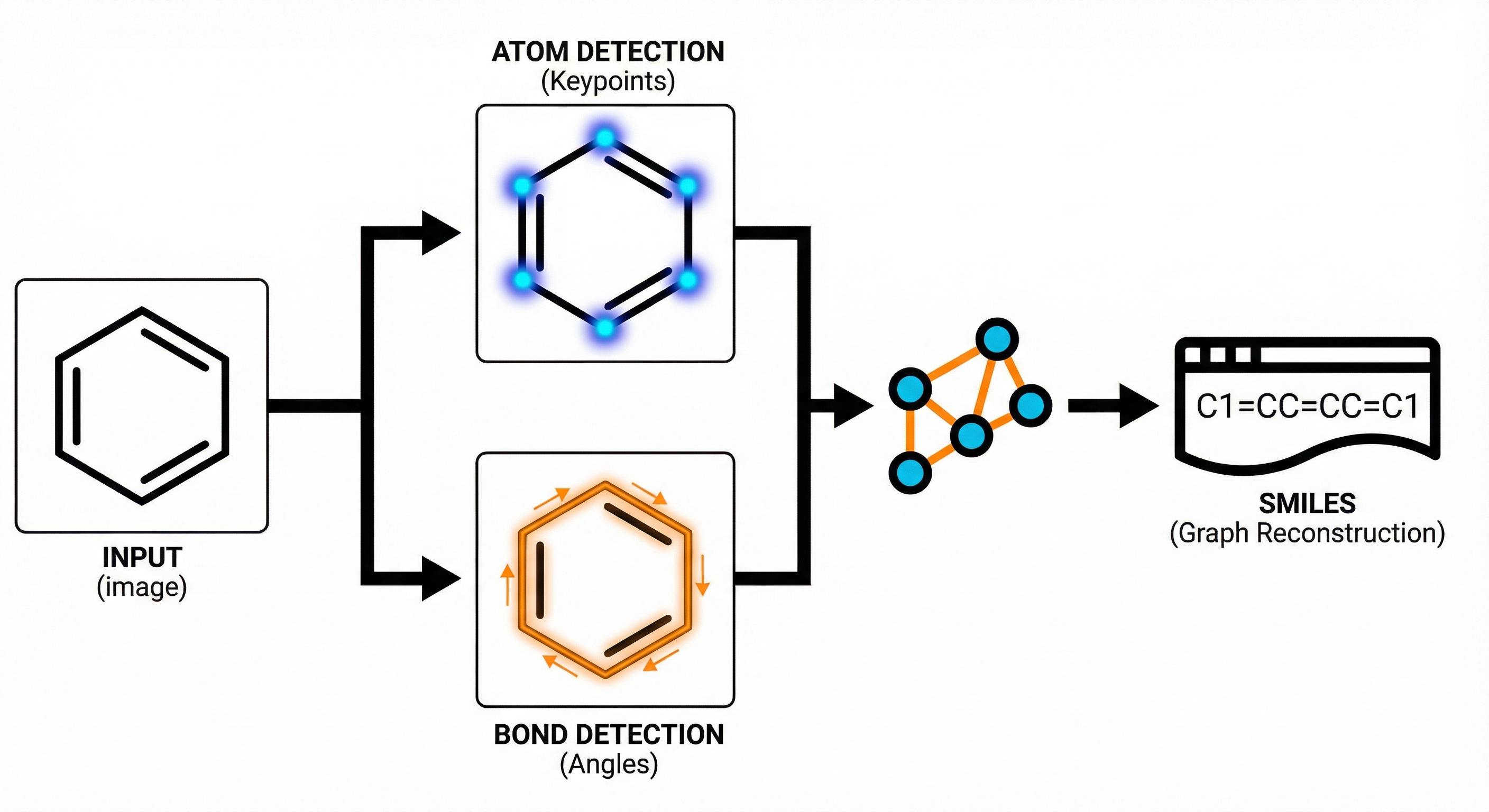
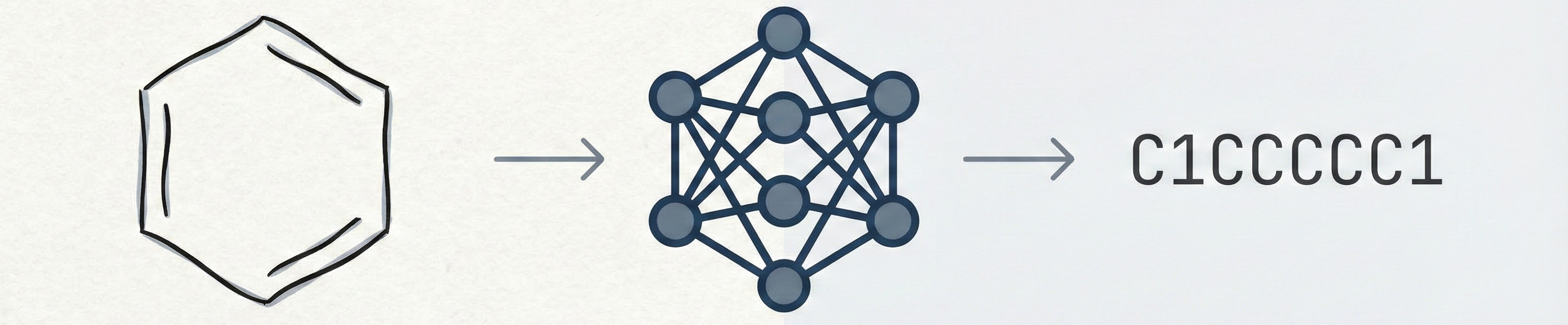
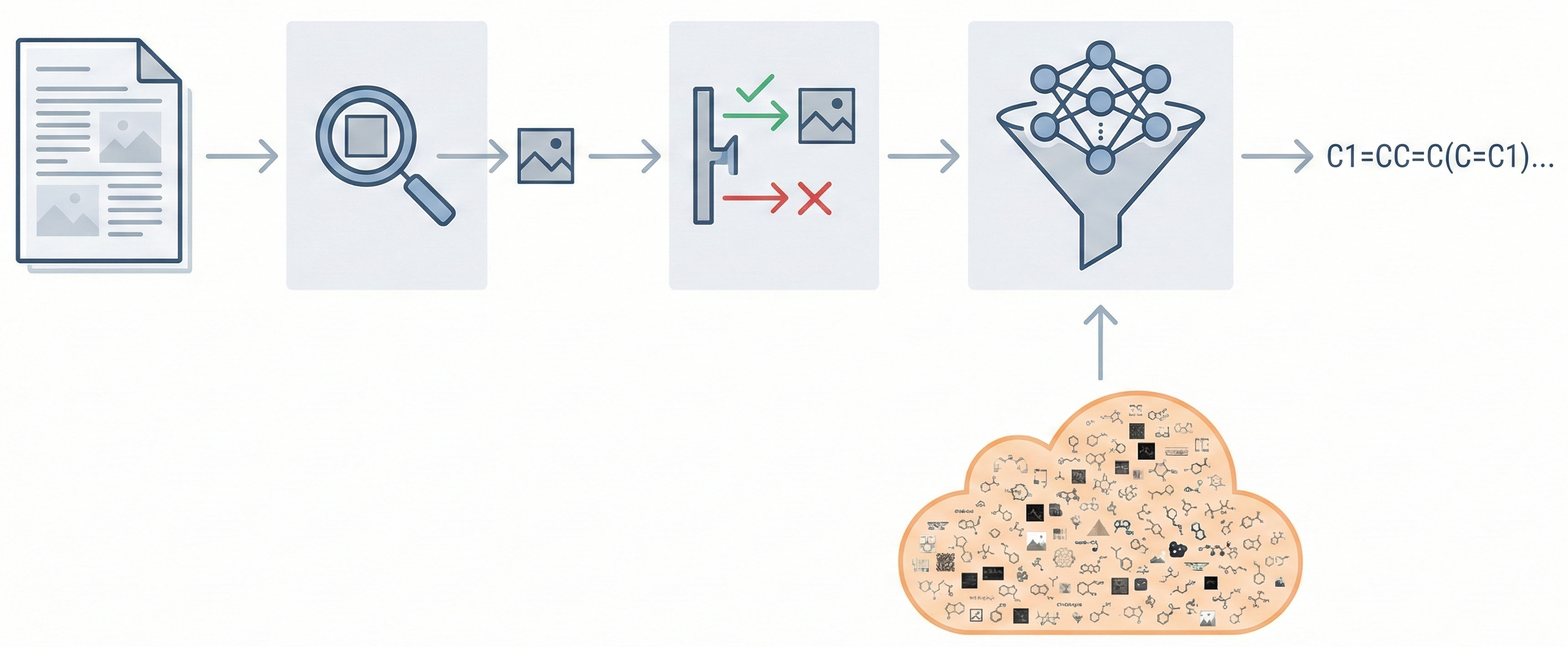
DECIMER.ai addresses the lack of open tools for Optical Chemical Structure Recognition (OCSR) by providing a comprehensive, deep-learning-based workflow. It features a novel data generation pipeline (RanDepict), a web application, and models for segmentation and recognition that rival or exceed proprietary solutions.