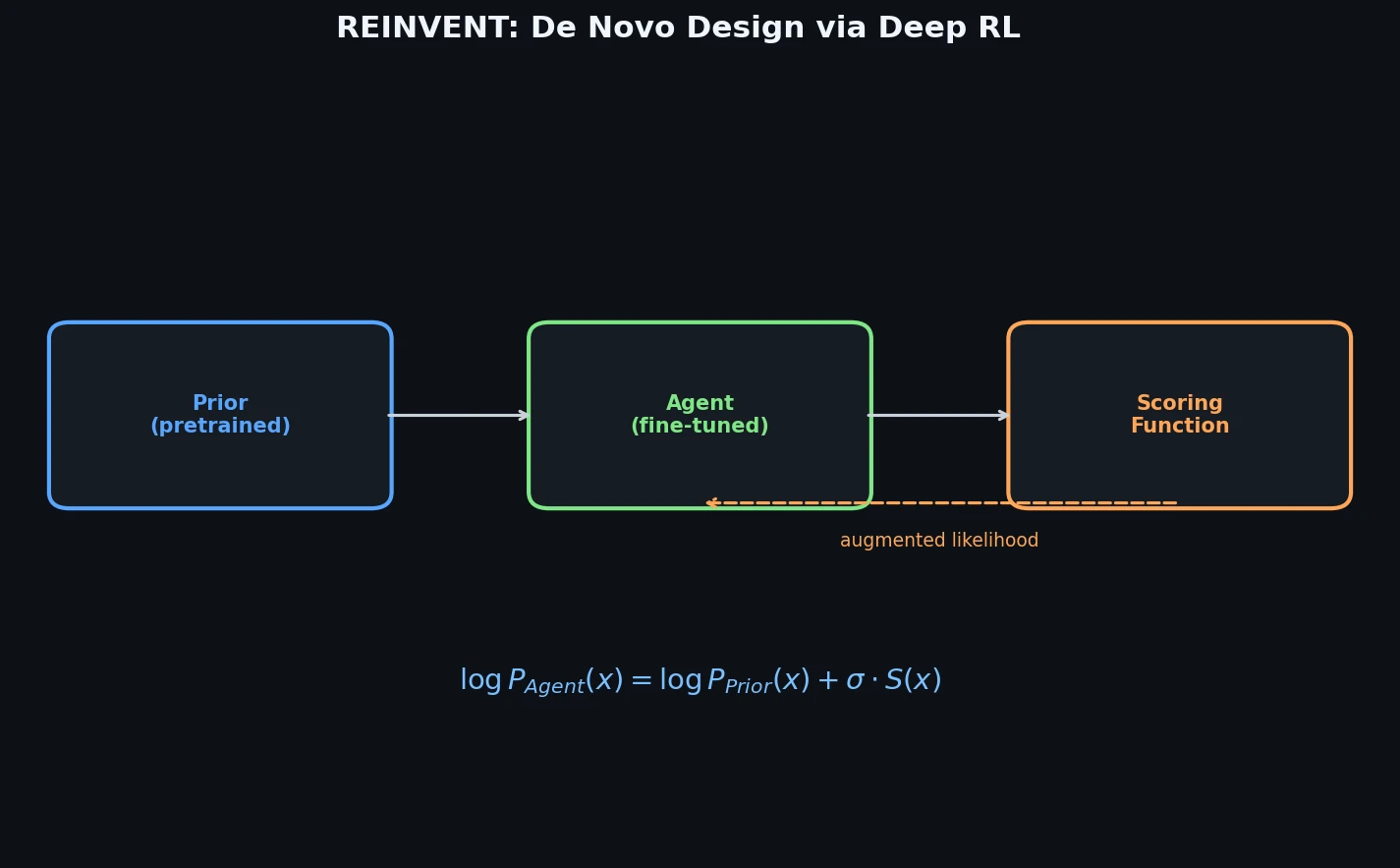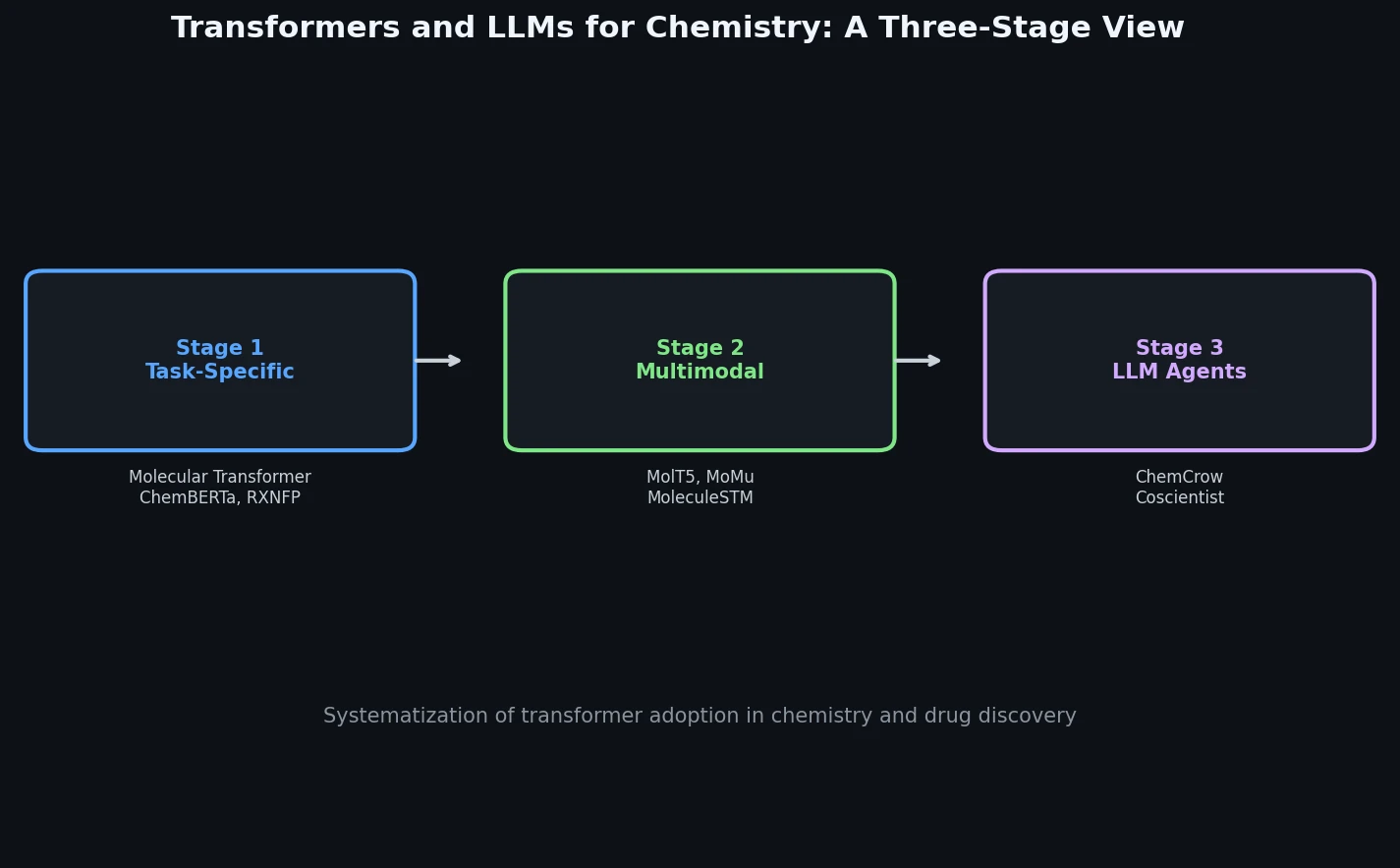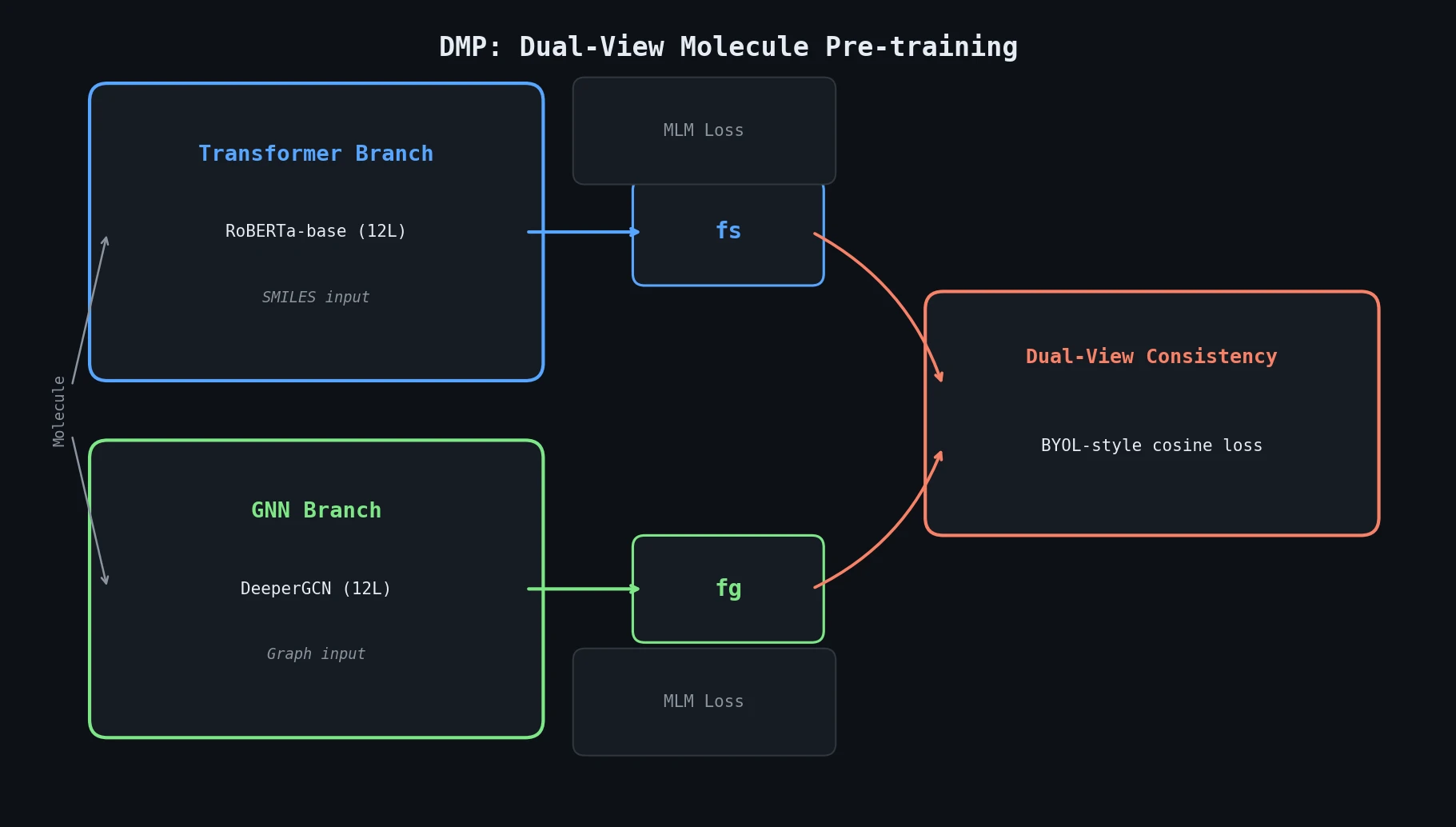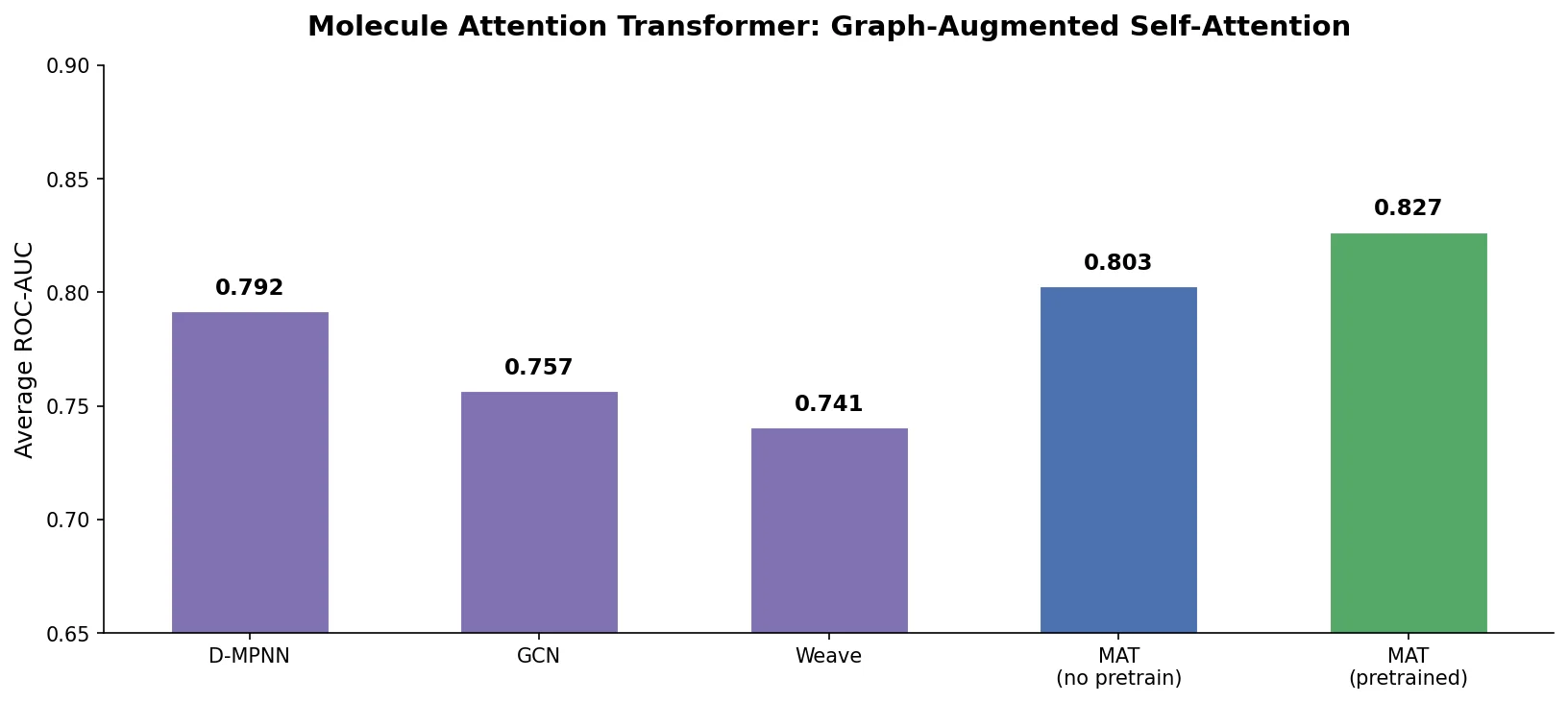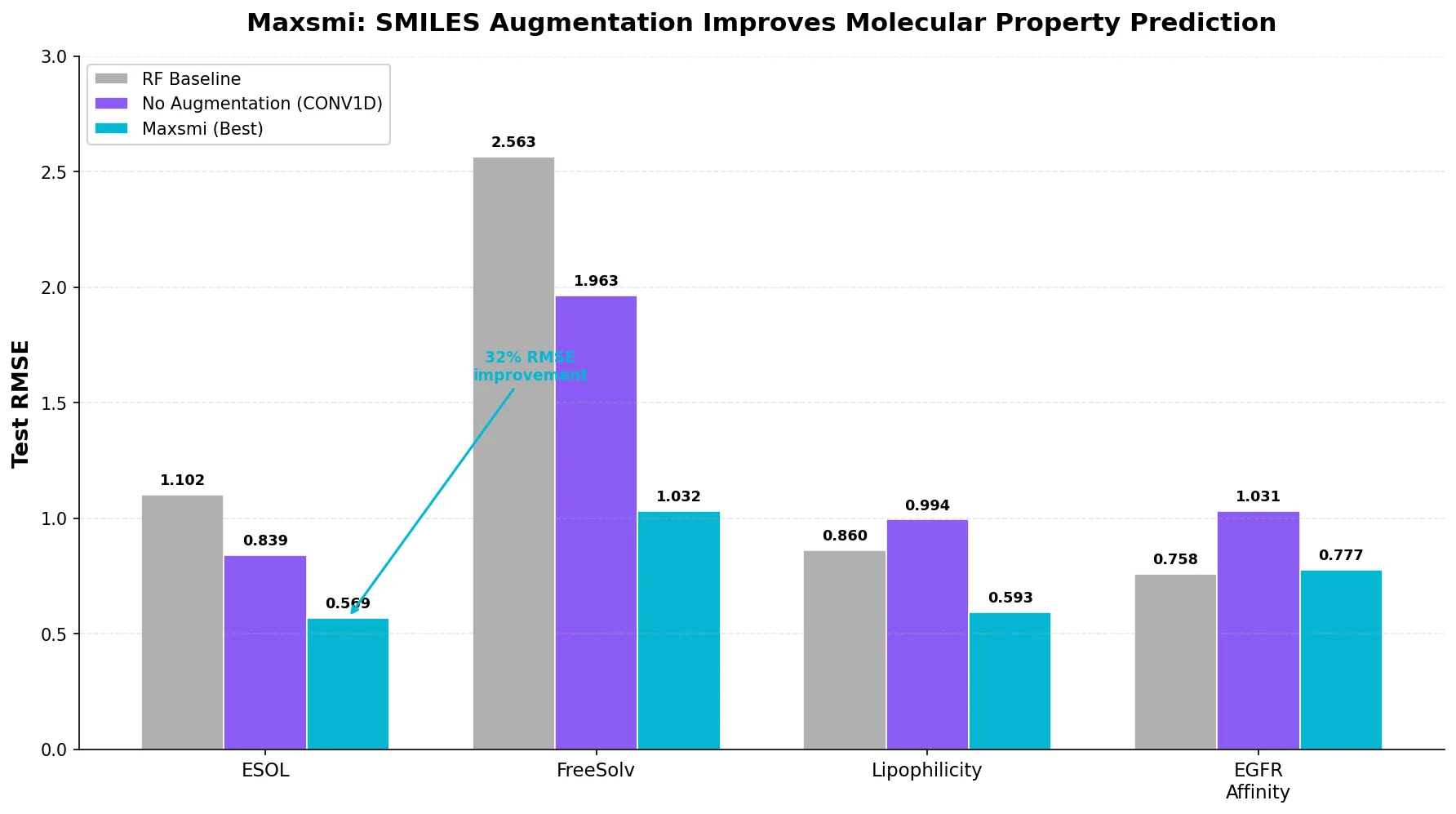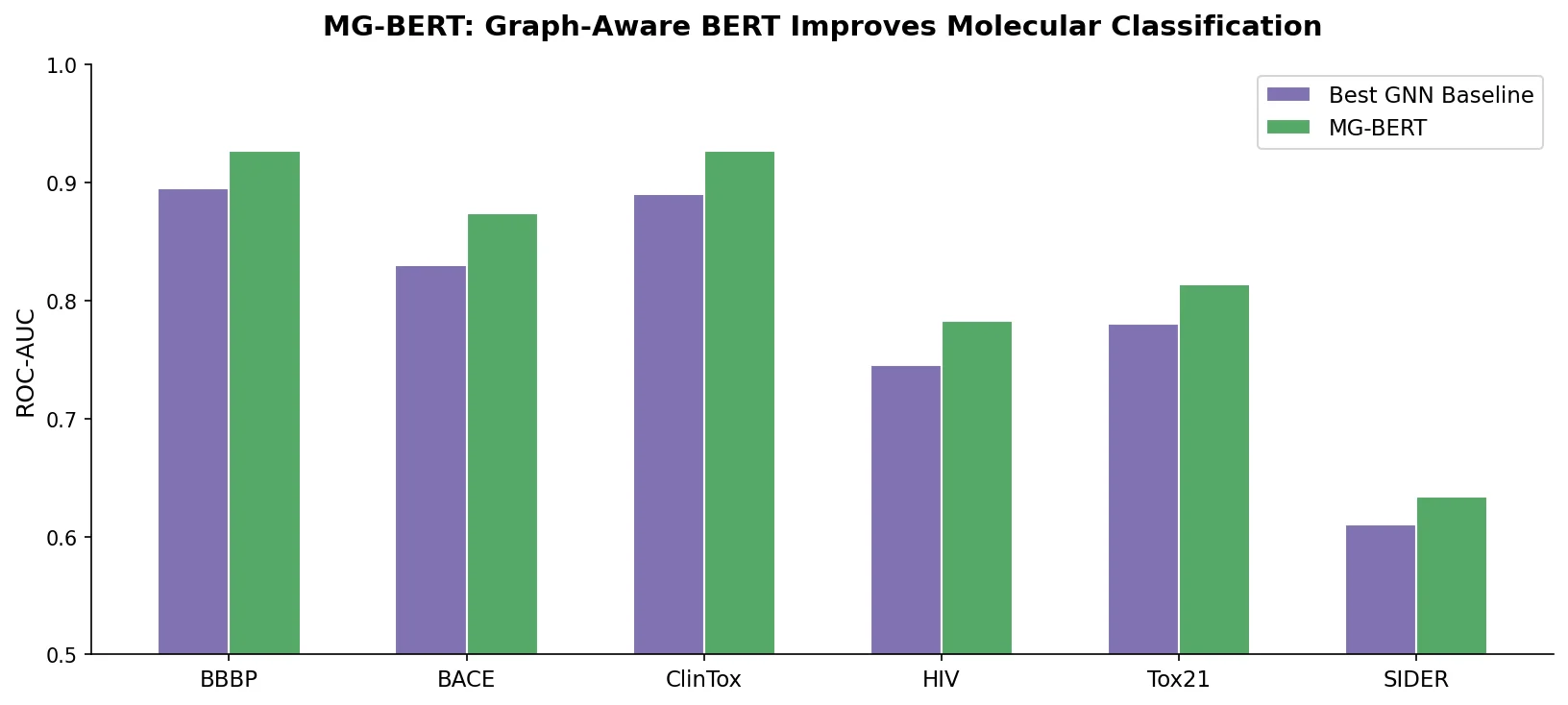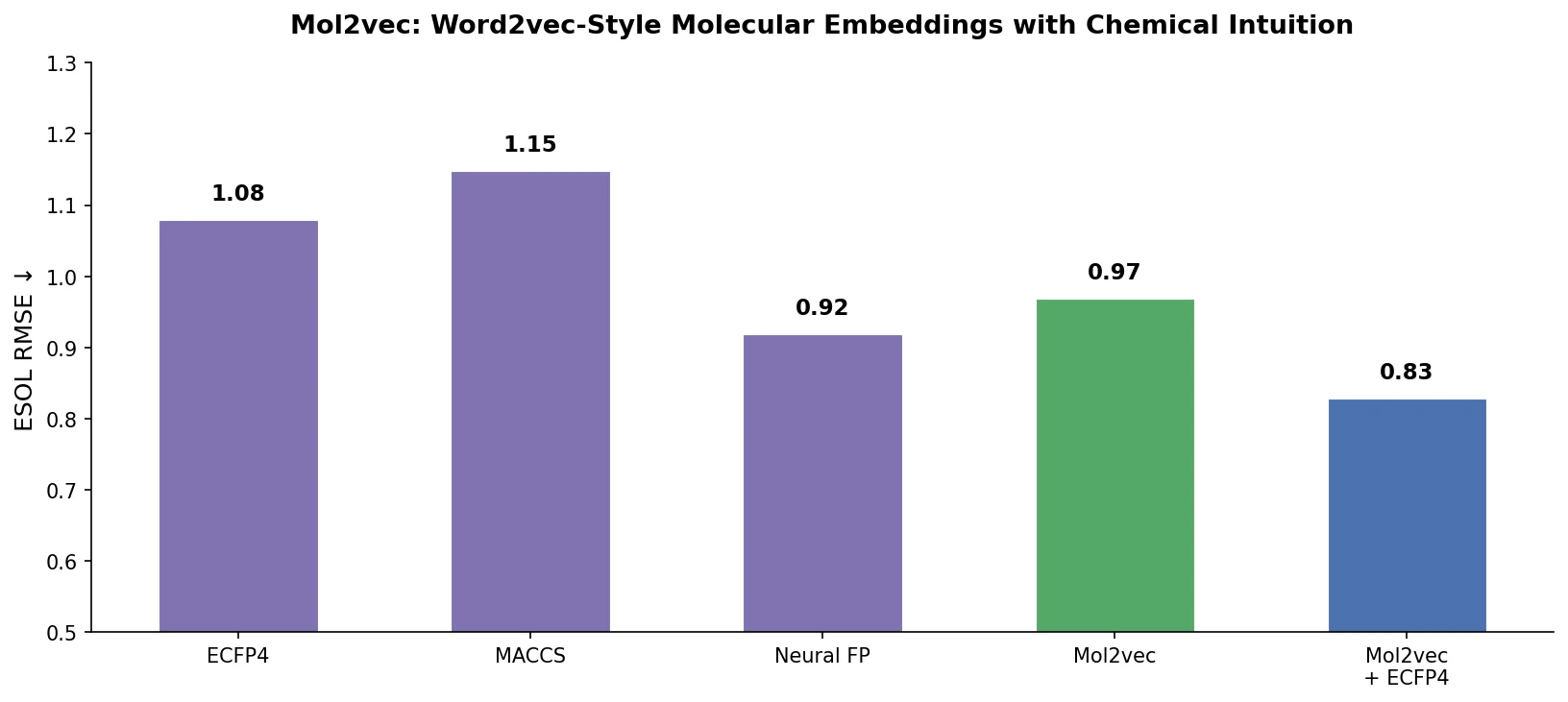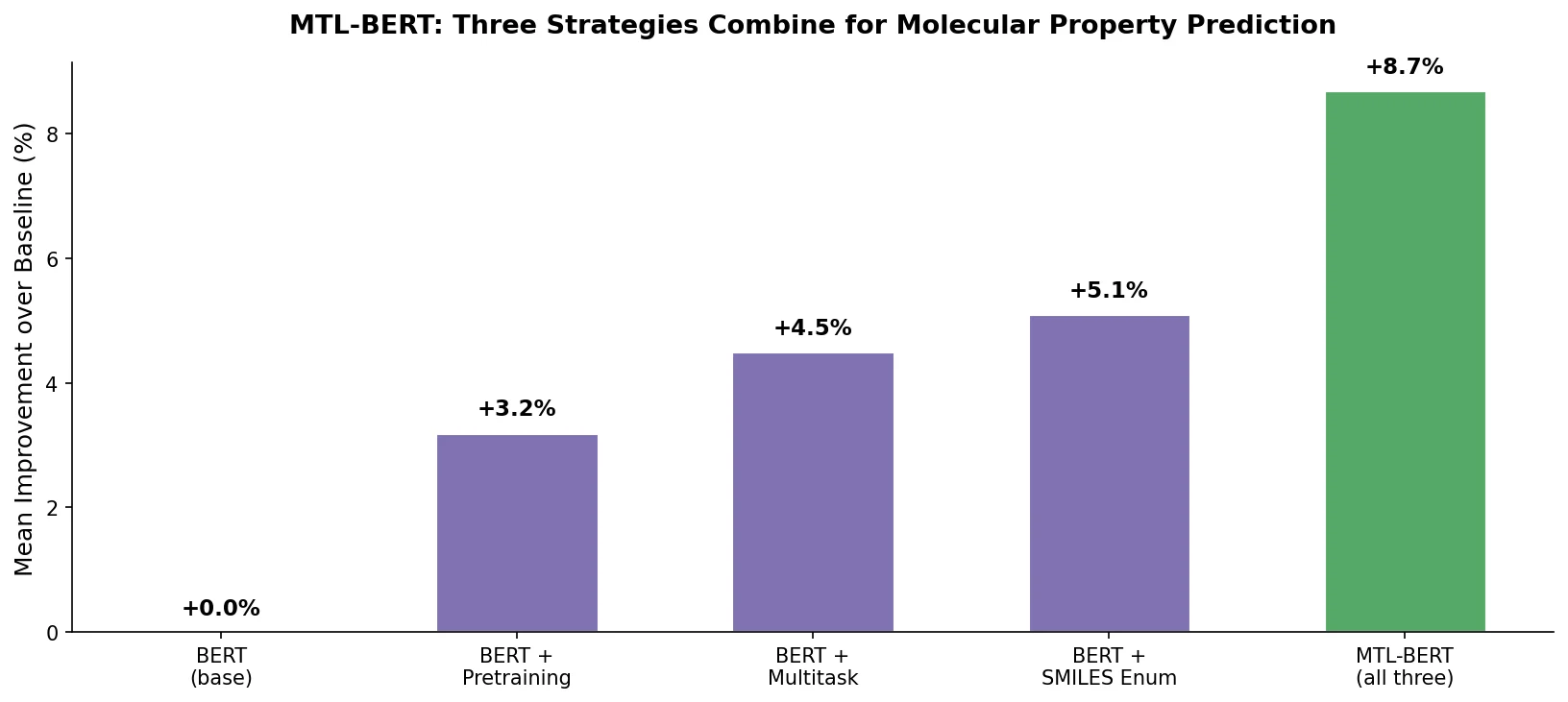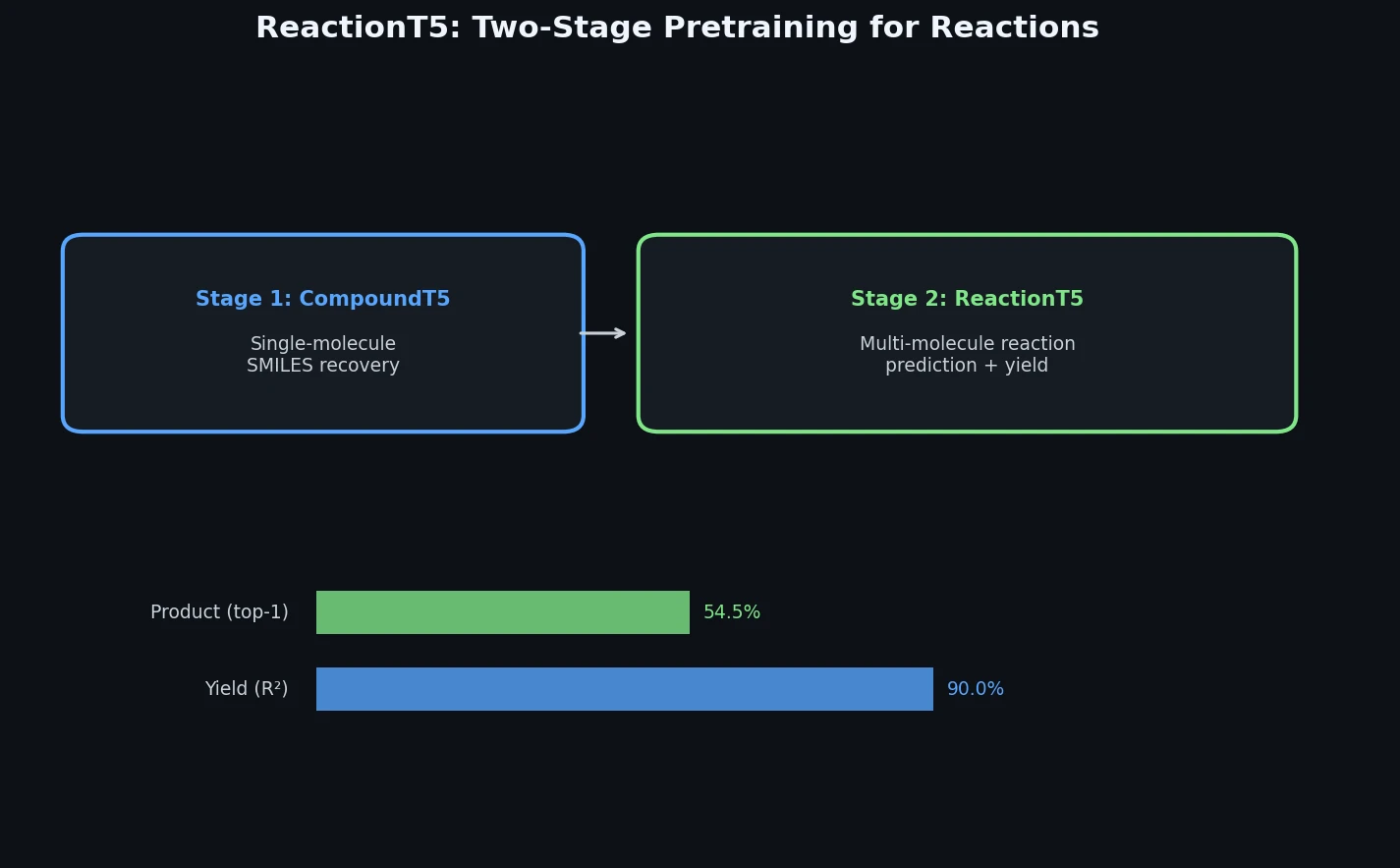
ReactionT5: Pre-trained T5 for Reaction Prediction
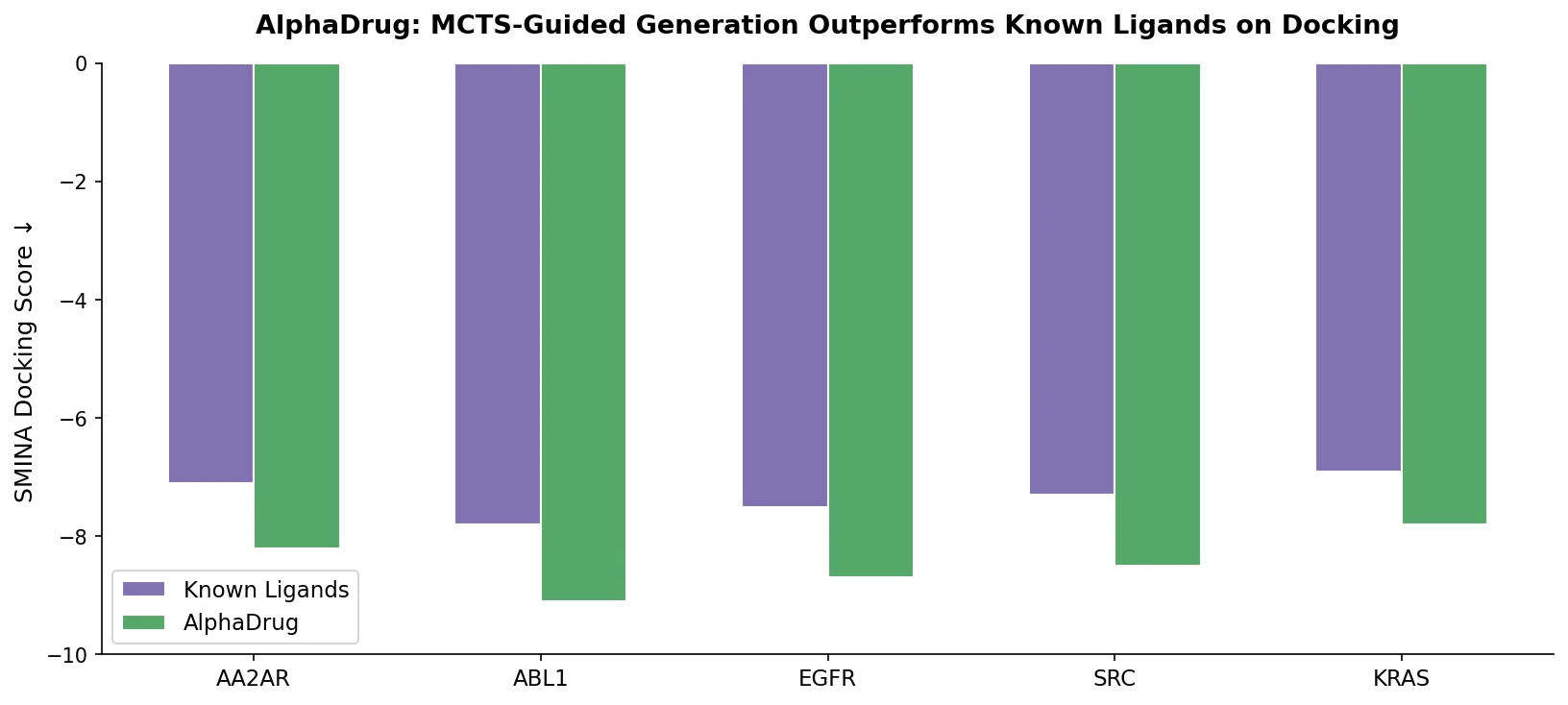
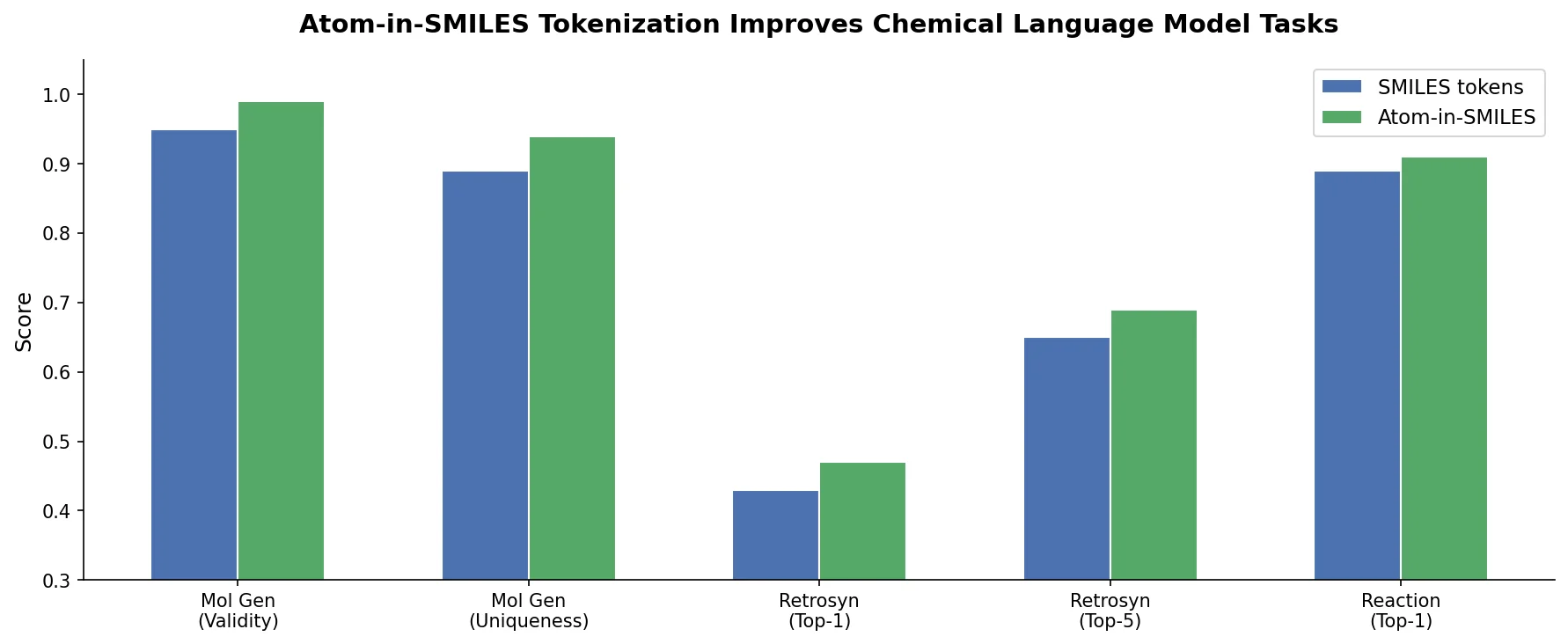
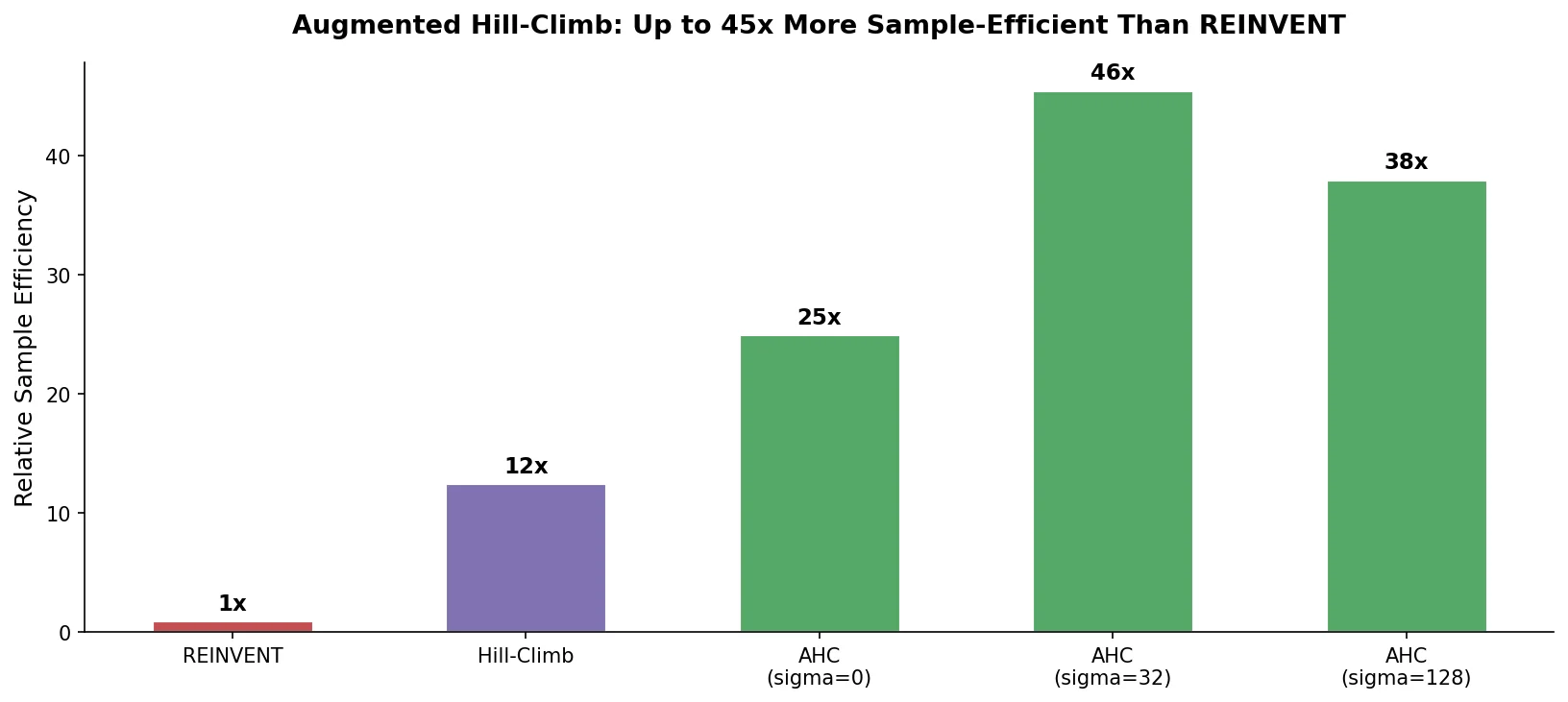
ReactionT5 introduces a two-stage pretraining pipeline (compound then reaction) on the Open Reaction Database, enabling competitive product and yield prediction with as few as 30 fine-tuning reactions.