Paper Information
Citation: Hewahi, N., Nounou, M. N., Nassar, M. S., Abu-Hamad, M. I., & Abu-Hamad, H. I. (2008). Chemical Ring Handwritten Recognition Based on Neural Networks. Ubiquitous Computing and Communication Journal, 3(3).
Publication: Ubiquitous Computing and Communication Journal 2008
Contribution: Recognition Architecture for Heterocyclic Rings
This is a Method paper ($\Psi_{\text{Method}}$).
It proposes a specific algorithmic architecture (the “Classifier-Recognizer Approach”) to solve a pattern recognition problem. The rhetorical structure centers on defining three variations of a method, performing ablation-like comparisons between them (Whole Image vs. Lower Part), and demonstrating superior performance metrics (~94% accuracy) for the proposed technique.
Motivation: Enabling Sketch-Based Chemical Search
The authors identify a gap in existing OCR and handwriting recognition research, which typically focuses on alphanumeric characters or whole words.
- Missing Capability: Recognition of specific heterocyclic chemical rings (23 types) had not been performed previously.
- Practical Utility: Existing chemical search engines require text-based queries (names); this work enables “backward” search where a user can draw a ring to find its information.
- Educational/Professional Aid: Useful for chemistry departments and mobile applications where chemists can sketch formulas on screens.
Innovation: The Classifier-Recognizer Pipeline
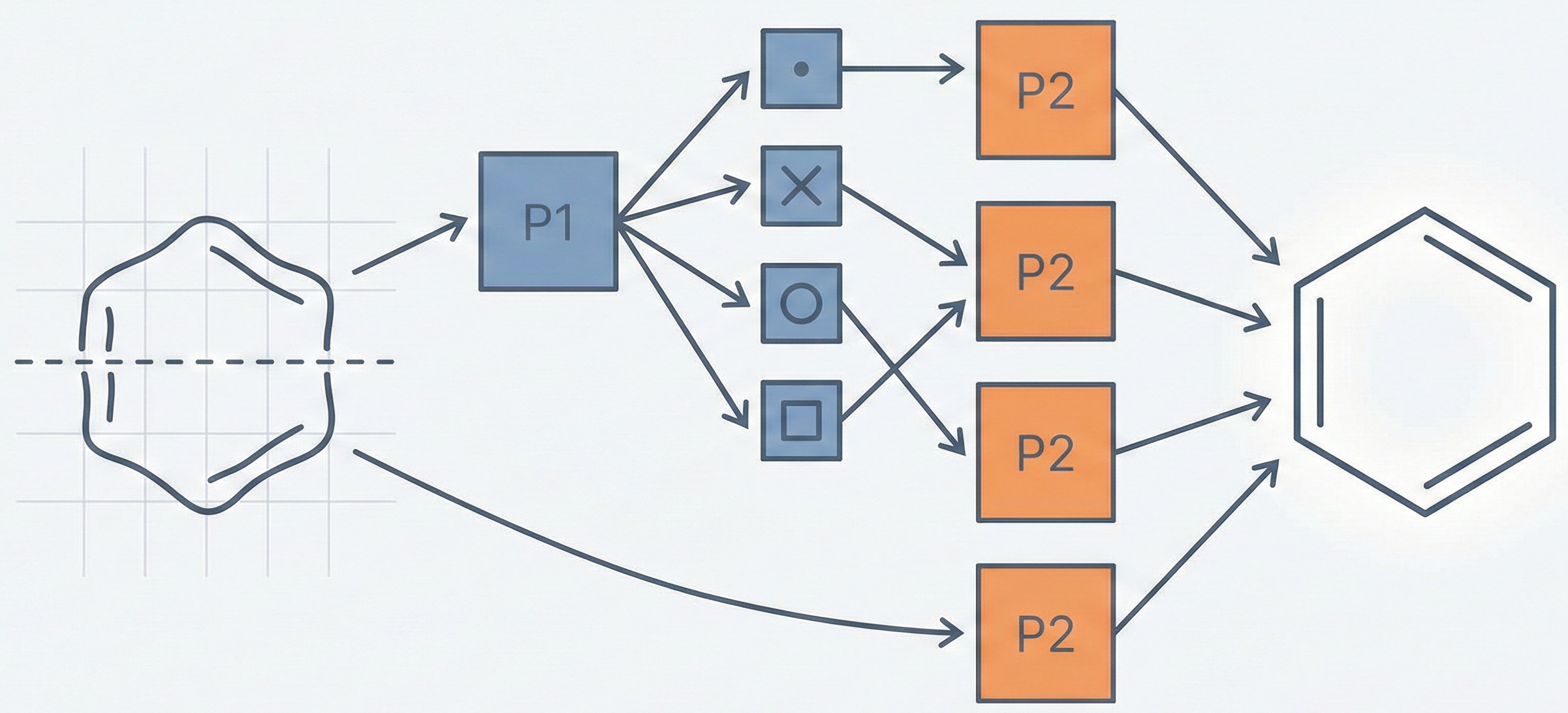
The core novelty is the two-phase “Classifier-Recognizer” architecture designed to handle the visual similarity of heterocyclic rings:
- Phase 1 (Classifier): A neural network classifies the ring into one of four broad categories (S, N, O, Others) based solely on the upper part of the image (40x15 pixels).
- Phase 2 (Recognizer): A class-specific neural network identifies the exact ring.
- Optimization: The most successful variation (“Lower Part Image Recognizer with Half Size Grid”) uses only the lower part of the image and odd rows (half-grid) to reduce input dimensionality and computation time while improving accuracy. This effectively subsamples the input grid matrix $M \in \mathbb{R}^{H \times W}$ to a reduced matrix $M_{\text{sub}}$: $$ M_{\text{sub}} = { m_{i,j} \in M \mid i \text{ is odd} } $$
Failed Preliminary Approaches
Before arriving at the Classifier-Recognizer architecture, the authors tried three simpler methods that all failed:
- Ordinary NN: A single neural network with 1600 inputs (40x40 grid), 1600 hidden units, and 23 outputs. This standard approach achieved only 7% accuracy.
- Row/Column pixel counts: Using the number of black pixels per row and per column as features ($N_c + N_r$ inputs), which dramatically reduced dimensionality. This performed even worse, below 1% accuracy.
- Midline crossing count: Drawing a horizontal midline and counting the number of line crossings. This failed because the crossing count varies between writers for the same ring.
These failures motivated the two-phase Classifier-Recognizer design.
Experimental Setup and Network Variations
The authors conducted a comparative study of three methodological variations:
- Whole Image Recognizer: Uses the full image.
- Whole Image (Half Size Grid): Uses only odd rows ($20 \times 40$ pixels).
- Lower Part (Half Size Grid): Uses the lower part of the image with odd rows (the proposed method).
Setup:
- Dataset: 23 types of heterocyclic rings.
- Training: 1500 samples (distributed across S, N, O, and Others classes).
- Testing: 1150 samples.
- Metric: Recognition accuracy (Performance %) and Error %.
Results: High Accuracy via Dimension Reduction
- Superior Method: The “Lower Part Image Recognizer with Half Size Grid” achieved the best performance (~94% overall).
- High Classifier Accuracy: The first phase (classification into S/N/O/Other) achieves 100% accuracy for class S, 98.67% for O, 97.75% for N, and 97.67% for Others (Table 3).
- Class ‘Others’ Difficulty: The ‘Others’ class showed lower performance (~90-93%) compared to S/N/O due to the higher complexity and similarity of rings in that category.
- Efficiency: The half-grid approach reduced training time from ~53 hours (Whole Image) to ~35 hours (Lower Part Half Size Grid) while improving accuracy from 87% to 94%.
Training/Testing comparison across the three Classifier-Recognizer variations (Table 2):
| Method | Hidden Nodes | Iterations | Training Time (hrs) | Error | Performance |
|---|---|---|---|---|---|
| Whole Image | 50 | 1000 | ~53 | 13.0% | 87.0% |
| Whole Image (Half Grid) | 50 | 1000 | ~41 | 9.0% | 91.0% |
| Lower Part (Half Grid) | 50 | 1000 | ~35 | 6.0% | 94.0% |
Reproducibility Details
Data
The dataset consists of handwritten samples of 23 specific heterocyclic rings.
| Purpose | Dataset | Size | Notes |
|---|---|---|---|
| Training | Heterocyclic Rings | 1500 samples | Split: 300 (S), 400 (N), 400 (O), 400 (Others) |
| Testing | Heterocyclic Rings | 1150 samples | Split: 150 (S), 300 (O), 400 (N), 300 (Others) |
Preprocessing Steps:
- Monochrome Conversion: Convert image to monochrome bitmap.
- Grid Scaling: Convert drawing area (regardless of original size) to a fixed 40x40 grid.
- Bounding: Scale the ring shape itself to fit the 40x40 grid.
Algorithms
The “Lower Part with Half Size” Pipeline:
- Cut Point: A horizontal midline is defined; the algorithm separates the “Upper Part” and “Lower Part”.
- Phase 1 Input: The Upper Part (rows 0-15 approx, scaled) is fed to the Classifier NN to determine the class (S, N, O, or Others).
- Phase 2 Input:
- For classes S, N, O: The Lower Part of the image is used.
- For class Others: The Whole Ring is used.
- Dimensionality Reduction: For the recognizer networks, only odd rows are used (effectively a 20x40 input grid) to reduce inputs from 1600 to 800.
Models
The system uses multiple distinct Feed-Forward Neural Networks (Backpropagation is implied by “training” and “epochs” context, though not explicitly named as the algorithm):
- Structure: 1 Classifier NN + 4 Recognizer NNs (one for each class).
- Hidden Layers: The preliminary “ordinary method” experiment used 1600 hidden units. The Classifier-Recognizer methods all used 50 hidden nodes per Table 2. The paper also notes that the ordinary approach tried various hidden layer sizes.
- Input Nodes:
- Standard: 1600 (40x40).
- Optimized: ~800 (20x40 via half-grid).
Evaluation
Classifier Phase Testing Results (Table 3):
| Class | Samples | Correct | Accuracy | Error |
|---|---|---|---|---|
| S | 150 | 150 | 100.00% | 0.00% |
| O | 300 | 296 | 98.67% | 1.33% |
| N | 400 | 391 | 97.75% | 2.25% |
| Others | 300 | 293 | 97.67% | 2.33% |
Recognizer Phase Testing Results (Lower Part Image Recognizer with Half Size Grid, Table 4):
| Class | Samples | Correct | Accuracy | Error |
|---|---|---|---|---|
| S | 150 | 147 | 98.00% | 2.00% |
| O | 300 | 289 | 96.33% | 3.67% |
| N | 400 | 386 | 96.50% | 3.50% |
| Others | 300 | 279 | 93.00% | 7.00% |
| Overall | 1150 | - | ~94.0% | - |
Reproducibility Assessment
No source code, trained models, or datasets were released with this paper. The handwritten ring samples were collected by the authors, and the software described (a desktop application) is not publicly available. The neural network architecture details (50 hidden nodes, 1000 iterations) and preprocessing pipeline are described in sufficient detail for reimplementation, but reproducing results would require collecting a new handwritten dataset of heterocyclic rings.
Status: Closed (no public code, data, or models).
Citation
@article{hewahiCHEMICALRINGHANDWRITTEN2008,
title = {CHEMICAL RING HANDWRITTEN RECOGNITION BASED ON NEURAL NETWORKS},
author = {Hewahi, Nabil and Nounou, Mohamed N and Nassar, Mohamed S and Abu-Hamad, Mohamed I and Abu-Hamad, Husam I},
year = {2008},
journal = {Ubiquitous Computing and Communication Journal},
volume = {3},
number = {3}
}