Paper Information
Citation: Ouyang, H., Liu, W., Tao, J., et al. (2024). ChemReco: automated recognition of hand-drawn carbon-hydrogen-oxygen structures using deep learning. Scientific Reports, 14, 17126. https://doi.org/10.1038/s41598-024-67496-7
Publication: Scientific Reports 2024
Additional Resources:
Research Contribution & Classification
This is a Methodological Paper ($\Psi_{\text{Method}}$) with a significant Resource ($\Psi_{\text{Resource}}$) component.
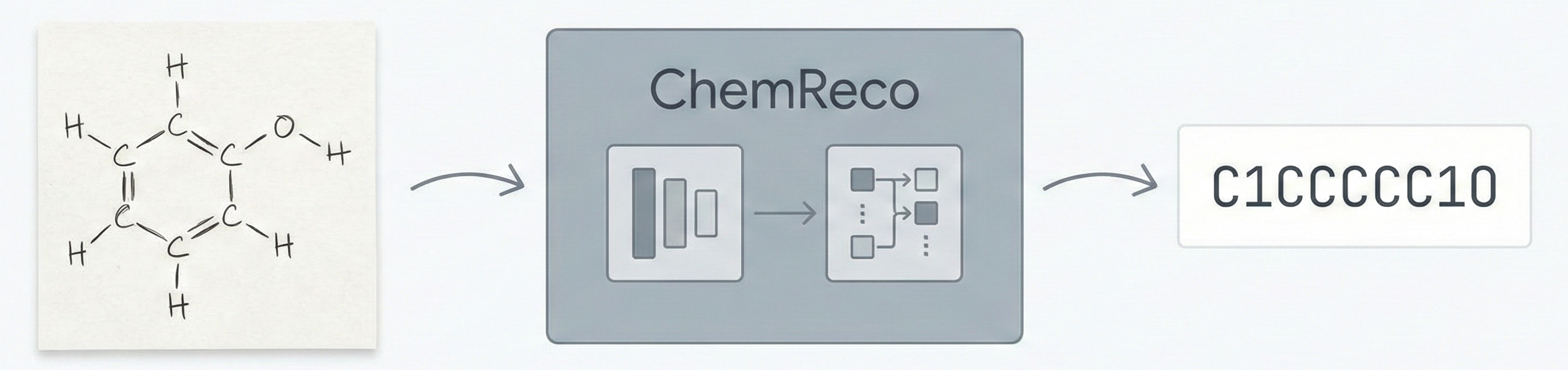
- Method: The primary contribution is “ChemReco,” a specific deep learning pipeline (EfficientNet + Transformer) designed to solve the Optical Chemical Structure Recognition (OCSR) task for hand-drawn images. The authors conduct extensive ablation studies on architecture and data mixing ratios to validate performance.
- Resource: The authors explicitly state that “the primary focus of this paper is constructing datasets” due to the scarcity of hand-drawn molecular data. They introduce a comprehensive synthetic data generation pipeline involving RDKit modifications and image degradation to create training data.
Motivation: Digitizing Hand-Drawn Chemical Sketches
Hand-drawing is the most intuitive method for chemists and students to record molecular structures. However, digitizing these drawings into machine-readable formats (like SMILES) usually requires time-consuming manual entry or specialized software.
- Gap: Existing OCSR tools and rule-based methods often fail on hand-drawn sketches due to diverse writing styles, poor image quality, and the absence of labeled data.
- Application: Automated recognition enables efficient chemical research and allows for automatic grading in educational settings.
Core Innovation: Synthetic Pipeline and Hybrid Architecture
The paper introduces ChemReco, an end-to-end system for recognizing C-H-O structures. Key novelties include:
- Synthetic Data Pipeline: A multi-stage generation method that modifies RDKit source code to randomize bond/angle parameters, followed by OpenCV-based augmentation, degradation, and background addition to simulate realistic hand-drawn artifacts.
- Architectural Choice: The specific application of EfficientNet (encoder) combined with a Transformer (decoder) for this domain, which the authors demonstrate outperforms the more common ResNet+LSTM baselines.
- Hybrid Training Strategy: Finding that a mix of 90% synthetic and 10% real data yields optimal performance, superior to using either dataset alone.
Methodology & Ablation Studies
The authors performed a series of ablation studies and comparisons:
- Synthesis Ablation: Evaluated the impact of each step in the generation pipeline (RDKit only $\rightarrow$ Augmentation $\rightarrow$ Degradation $\rightarrow$ Background) on validation loss and accuracy.
- Dataset Size Ablation: Tested model performance when trained on synthetic datasets ranging from 100,000 to 1,000,000 images.
- Real/Synthetic Ratio: Investigated the optimal mixing ratio of synthetic to real hand-drawn images (100:0, 90:10, 50:50, 10:90, 0:100), finding that the 90:10 ratio achieved 93.81% exact match, compared to 63.33% for synthetic-only and 65.83% for real-only.
- Architecture Comparison: Benchmarked four encoder-decoder combinations: ResNet vs. EfficientNet encoders paired with LSTM vs. Transformer decoders.
- Baseline Comparison: Compared results against a related study utilizing a CNN+LSTM framework.
Results & Interpretations
- Best Performance: The EfficientNet + Transformer model trained on a 90:10 synthetic-to-real ratio achieved a 96.90% Exact Match rate on the test set.
- Background Robustness: When training on synthetic data alone (no real images), the best accuracy on background-free test images was approximately 46% (using RDKit-aug-deg), while background test images reached approximately 53% (using RDKit-aug-bkg-deg). Adding random backgrounds during training helped prevent the model from overfitting to clean white backgrounds.
- Data Volume: Increasing the synthetic dataset size from 100k to 1M consistently improved accuracy (average exact match: 49.40% at 100k, 54.29% at 200k, 61.31% at 500k, 63.33% at 1M, all without real images in training).
- Encoder-Decoder Comparison (at 90:10 mix with 1M images):
| Encoder | Decoder | Avg. Exact Match (%) |
|---|---|---|
| ResNet | LSTM | 93.81 |
| ResNet | Transformer | 94.76 |
| EfficientNet | LSTM | 96.31 |
| EfficientNet | Transformer | 96.90 |
- Superiority over Baselines: The model outperformed the cited CNN+LSTM baseline from ChemPix (93% vs 76% on the ChemPix test set).
Limitations
- Restricted atom types: The system only handles molecules composed of carbon, hydrogen, and oxygen (C-H-O), excluding nitrogen, sulfur, halogens, and other heteroatoms commonly found in organic chemistry.
- Structural complexity: Only structures with at most one ring are supported. Complex multi-ring systems and fused ring structures are not covered.
- Dataset availability: The real hand-drawn dataset (2,598 images) is not publicly released and is only available upon request from the corresponding author.
- Future directions: The authors suggest expanding to more heteroatoms, complex ring structures, and applications in automated grading of chemistry exams.
Reproducibility
| Artifact | Type | License | Notes |
|---|---|---|---|
| hdr-DeepLearning | Code | Unknown | Official implementation in PyTorch |
| Paper | Publication | CC-BY-4.0 | Open access via Nature |
The real hand-drawn dataset (2,598 images) is available upon request from the corresponding author, not publicly downloadable. The synthetic data generation pipeline is described in detail but relies on modified RDKit source code, which is included in the repository.
Data
The study utilizes a combination of collected SMILES data, real hand-drawn images, and generated synthetic images.
- Source Data: SMILES codes collected from PubChem, ZINC, GDB-11, and GDB-13. Filtered for C, H, O atoms and max 1 ring.
- Real Dataset: 670 selected SMILES codes drawn by multiple volunteers, totaling 2,598 images.
- Synthetic Dataset: Generated up to 1,000,000 images using the pipeline below.
- Training Mix: The optimal training set used 1 million images with a 90:10 ratio of synthetic to real images.
| Dataset Type | Source | Size | Notes |
|---|---|---|---|
| Real | Volunteer Drawings | 2,598 images | Used for mixed training and testing |
| Synthetic | Generated | 100k - 1M | Generated via modified RDKit + OpenCV augmentation/degradation; optionally enhanced with Stable Diffusion |
Algorithms
The Synthetic Image Generation Pipeline is critical for reproduction:
- RDKit Modification: Modify source code to introduce random keys, character width, length, and bond angles.
- Augmentation (OpenCV): Apply sequence: Resize ($p=0.5$), Blur ($p=0.4$), Erode/Dilate ($p=0.2$), Distort ($p=0.8$), Flip ($p=0.5$), Affine ($p=0.7$).
- Degradation: Apply sequence: Salt+pepper noise ($p=0.1$), Contrast ($p=0.7$), Sharpness ($p=0.5$), Invert ($p=0.3$).
- Background Addition: Random backgrounds are augmented (Crop, Distort, Flip) and added to the molecular image to prevent background overfitting.
- Diffusion Enhancement: Stable Diffusion (v1-4) is used for image-to-image enhancement to better simulate hand-drawn styles (prompt: “A pencil sketch of [Formula]… without charge distribution”).
Models
The system uses an encoder-decoder architecture:
- Encoder: EfficientNet (pre-trained on ImageNet). The last layer is removed, and features are extracted into a Numpy array.
- Decoder: Transformer. Utilizes self-attention to generate the SMILES sequence. Chosen over LSTM for better handling of long-range dependencies.
- Output: Canonical SMILES string.
Evaluation
- Primary Metric: Exact Match (EM). A strict binary evaluation checking whether the complete generated SMILES perfectly replicates the target string.
- Other Metrics: Levenshtein Distance measures edit-level character proximity, while the Tanimoto coefficient evaluates structural similarity based on chemical fingerprints. Both were monitored during validation ablation runs.
| Metric | Value | Baseline (CNN+LSTM) | Notes |
|---|---|---|---|
| Exact Match | 96.90% | 76% | Tested on the provided test set |
Hardware
- CPU: Intel(R) Xeon(R) Gold 6130 (40 GB RAM).
- GPU: NVIDIA Tesla V100 (32 GB video memory).
- Framework: PyTorch 1.9.1.
- Training Configuration:
- Optimizer: Adam (learning rate 1e-4).
- Batch size: 32.
- Epochs: 100.
Citation
@article{ouyangChemRecoAutomatedRecognition2024,
title = {ChemReco: Automated Recognition of Hand-Drawn Carbon--Hydrogen--Oxygen Structures Using Deep Learning},
author = {Ouyang, Hengjie and Liu, Wei and Tao, Jiajun and Luo, Yanghong and Zhang, Wanjia and Zhou, Jiayu and Geng, Shuqi and Zhang, Chengpeng},
journal = {Scientific Reports},
volume = {14},
number = {1},
pages = {17126},
year = {2024},
publisher = {Nature Publishing Group},
doi = {10.1038/s41598-024-67496-7}
}