Paper Information
Citation: Rajan, K., Zielesny, A. & Steinbeck, C. (2020). DECIMER: towards deep learning for chemical image recognition. Journal of Cheminformatics, 12(65). https://doi.org/10.1186/s13321-020-00469-w
Publication: Journal of Cheminformatics 2020
Additional Resources:
Contribution: Method for Optical Chemical Entity Recognition
This is primarily a Method ($\Psi_{\text{Method}}$) paper with a strong Resource ($\Psi_{\text{Resource}}$) component.
- Method: It proposes a novel architecture (DECIMER) that repurposes “show-and-tell” image captioning networks for Optical Chemical Entity Recognition (OCER), providing an alternative to traditional rule-based segmentation pipelines.
- Resource: It establishes a framework for generating massively scalable synthetic training data using open-source cheminformatics tools (CDK) and databases (PubChem), circumventing the scarcity of manually annotated chemical images.
Motivation: Brittleness of Heuristic Pipelines
The extraction of chemical structures from scientific literature (OCER) is critical for populating open-access databases. Traditional OCER systems (like OSRA or CLiDE) rely on complex multi-step pipelines involving vectorization, character recognition, and graph compilation. These systems are brittle and incorporating new structural features requires laborious engineering. Inspired by deep reinforcement learning systems like AlphaGo Zero, the authors sought to formulate an end-to-end deep learning approach that learns directly from data with minimal prior assumptions.
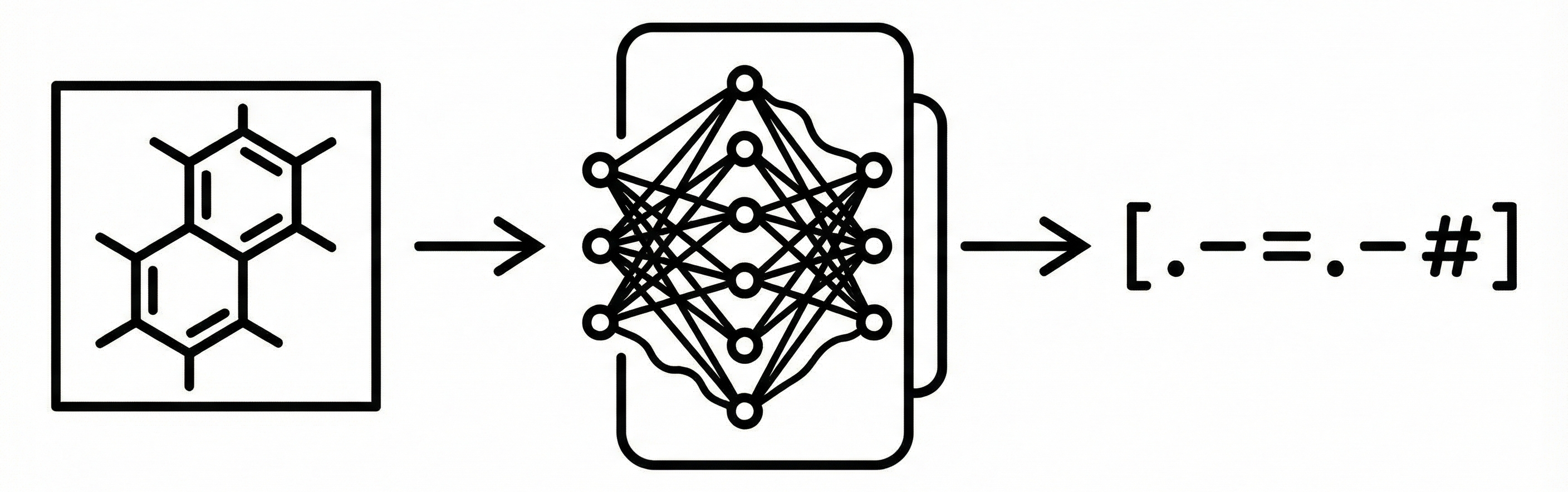
Novelty: Image Captioning for Molecular Graphs
- Image-to-Text Formulation: The paper frames chemical structure recognition as an image captioning problem, translating a bitmap image directly into a SMILES string using an encoder-decoder network. This bypasses explicit segmentation of atoms and bonds entirely.
- Synthetic Data Strategy: The authors generate synthetic images from PubChem using the CDK Structure Diagram Generator, scaling the dataset size to 15 million.
- Robust String Representations: The study performs key ablation experiments on string representations, comparing standard SMILES against DeepSMILES to evaluate how syntactic validity affects the network’s learning capability.
Experimental Setup and Validation Strategies
- Data Scaling: Models were trained on dataset sizes ranging from 54,000 to 15 million synthetic images to observe empirical scaling laws regarding accuracy and compute time.
- Representation Comparison: The authors compared the validity of predicted strings and recognition accuracy when training on SMILES versus DeepSMILES. The cross-entropy loss formulation for sequence generation can be represented as: $$ \mathcal{L} = -\sum_{t=1}^{T} \log P(y_t \mid y_{<t}, \mathbf{x}) $$ where $\mathbf{x}$ is the image representation and $y_t$ are the tokens of the SMILES/DeepSMILES string.
- Metric Evaluation: Performance was measured using Validity (syntactic correctness) and Tanimoto Similarity $T$, computed on molecular fingerprints to capture partial correctness even if the exact string prediction failed: $$ T(A, B) = \frac{|A \cap B|}{|A| + |B| - |A \cap B|} $$
Results and Critical Conclusions
- Data Representation: DeepSMILES proved superior to standard SMILES for training stability and output validity. Preliminary tests suggested SELFIES performs even better (0.78 Tanimoto vs 0.53 for DeepSMILES at 6M images).
- Scaling Behavior: Accuracy improves linearly with dataset size. The authors extrapolate that near-perfect detection would require training on 50 to 100 million structures.
- Current Limitations: At the reported training scale (up to 15M), the model does not yet rival traditional heuristic approaches, but the learning curve suggests it is a viable trajectory given sufficient compute and data.
Reproducibility Details
Data
The training data is synthetic, generated using the Chemistry Development Kit (CDK) Structure Diagram Generator (SDG) based on molecules from PubChem.
Curation Rules (applied to PubChem data):
- Molecular weight < 1500 Daltons.
- Elements restricted to: C, H, O, N, P, S, F, Cl, Br, I, Se, B.
- No counter ions or charged groups.
- No isotopes (e.g., D, T).
- Bond count between 5 and 40.
- SMILES length < 40 characters.
- Implicit hydrogens only (except in functional groups).
Preprocessing:
- Images: Generated as 299x299 bitmaps to match Inception V3 input requirements.
- Augmentation: One random rotation applied per molecule; no noise or blurring added in this iteration.
| Purpose | Dataset | Size | Notes |
|---|---|---|---|
| Training | Synthetic (PubChem) | 54k - 15M | Scaled across 12 experiments |
| Testing | Independent Set | 6k - 1.6M | 10% of training size |
Algorithms
- Architecture:
"Show, Attend and Tell"(Attention-based Image Captioning). - Optimization: Adam optimizer with learning rate 0.0005.
- Loss Function: Sparse Categorical Crossentropy.
- Training Loop: Trained for 25 epochs per model. Batch size of 640 images.
Models
The network is implemented in TensorFlow 2.0.
- Encoder: Inception V3 (Convolutional NN), used unaltered. Extracts feature vectors saved as NumPy arrays.
- Decoder: Gated Recurrent Unit (GRU) based Recurrent Neural Network (RNN) with soft attention mechanism.
- Embeddings: Image embedding dimension size of 600.
Evaluation
The primary metric is Tanimoto similarity (Jaccard index) on PubChem fingerprints, which is robust for measuring structural similarity even when exact identity is not reached.
| Metric | Definition |
|---|---|
| Tanimoto 1.0 | Percentage of predictions that are chemically identical to ground truth (isomorphic). |
| Average Tanimoto | Mean similarity score across the test set (captures partial correctness). |
| Validity | Percentage of predicted strings that are valid DeepSMILES/SMILES. |
Hardware
Training was performed on a single node.
- GPU: 1x NVIDIA Tesla V100.
- CPU: 2x Intel Xeon Gold 6230.
- RAM: 384 GB.
- Compute Time:
- Linear scaling with data size.
- 15 million structures took ~27 days (64,909s per epoch).
- Projected time for 100M structures: ~4 months on single GPU.
Citation
@article{rajanDECIMERDeepLearning2020,
title = {{{DECIMER}}: Towards Deep Learning for Chemical Image Recognition},
shorttitle = {{{DECIMER}}},
author = {Rajan, Kohulan and Zielesny, Achim and Steinbeck, Christoph},
year = 2020,
month = dec,
journal = {Journal of Cheminformatics},
volume = {12},
number = {1},
pages = {65},
issn = {1758-2946},
doi = {10.1186/s13321-020-00469-w}
}