Exhaustive Enumeration of Heteroaromatic Ring Systems
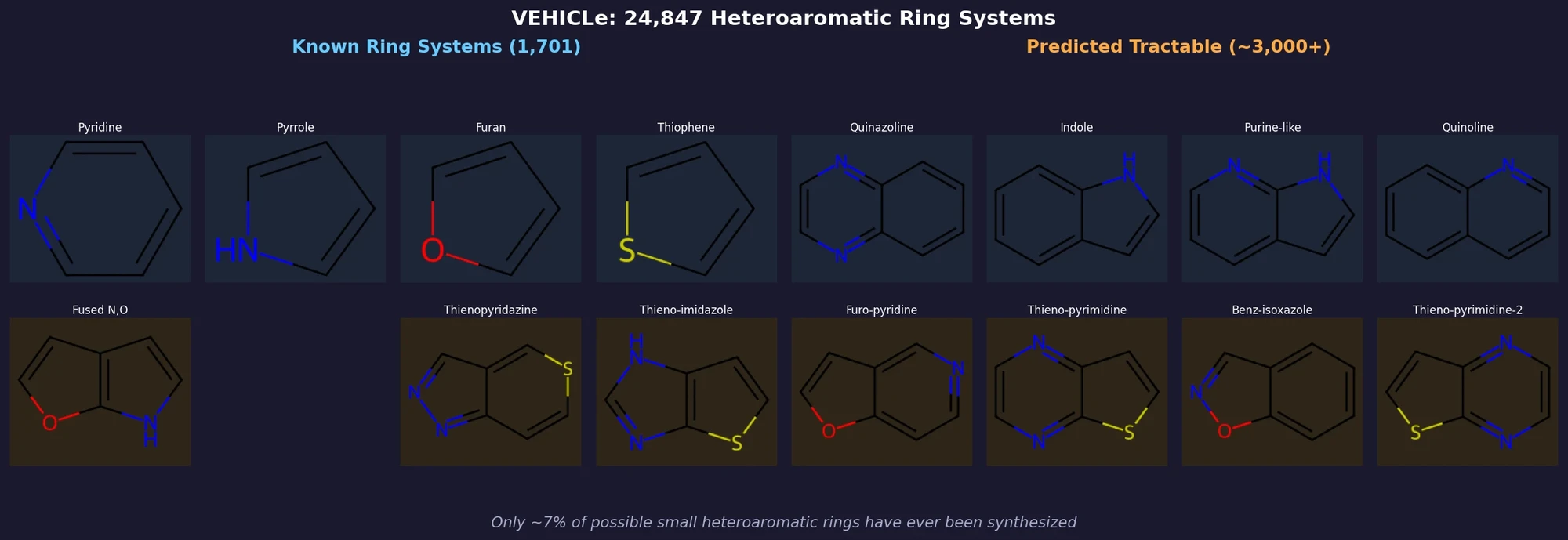
VEHICLe (Virtual Exploratory Heterocyclic Library) is a complete enumeration of all possible heteroaromatic ring systems under a set of constraints designed to capture the ring types most relevant to medicinal chemistry. The library contains 24,867 ring systems (23,895 after collapsing tautomers), yet only 1,701 of these have ever appeared in published compounds across databases totaling over 10 million molecules. The authors use this complete library to predict which unsynthesized ring systems could plausibly be made and to challenge organic chemists to conquer them.
Why Heteroaromatic Rings Matter for Drug Design
Heteroaromatic rings are central to synthetic bioactive small molecules for several reasons: they bind proteins efficiently through shape and hydrophobicity, their rigidity combined with heteroatom hydrogen bonding provides target selectivity, they support parallelizable coupling reactions (Suzuki, Stille) for rapid SAR exploration, multiple substitution positions can be explored without introducing stereocenters, and unusual ring systems or substitution patterns provide patent novelty. These advantages come with tradeoffs: low aqueous solubility, restricted SAR from rigidity, tendency toward molecular bloat during optimization, and difficulty achieving patent novelty with well-explored ring systems.
VEHICLe Construction
The library is built through a simple combinatorial pipeline implemented in Pipeline Pilot (Accelrys Software Inc.) that runs in about 3 minutes on a single-core 3 GHz Intel Xeon workstation:
- Building blocks: Six atomic units (C, N, O, S variants with appropriate bond types) serve as starting materials.
- Chain formation: Building blocks are combined into all possible chains of length 5 and 6 using two bond-forming rules (single and double bond).
- Ring closure: Chains are closed into five- and six-membered rings using three closure rules. Only rings satisfying Hückel’s $4n + 2$ aromaticity rule are retained.
- Ring fusion: Monocyclic rings are fused pairwise into all possible bicyclic combinations using four fusion rules. Aromatic bicycles are retained.
The enumeration constraints are: mono- and bicyclic rings only, five- and six-membered rings only, atoms restricted to C, N, O, S, and H, all neutral, all aromatic by Hückel’s rule, and only exocyclic carbonyls allowed. Including the carbonyl building block expands the library from 2,986 to 24,867 ring systems. Within this count, 1,744 tautomeric pairs exist in 772 clusters. Building blocks are input as MDL mol files, chains formed using MDL REACCS rxn format reactions, and duplicates removed by canonical SMILES comparison.
The following table summarizes VEHICLe ring system coverage across the compound datasets used for analysis:
| Dataset | Molecules | Distinct Ring Systems | VEHICLe Rings | VEHICLe % |
|---|---|---|---|---|
| Launched + Phases II/III | 2,461 | 950 | 120 | 13% |
| Phase I | 730 | 494 | 86 | 17% |
| Derwent patents | 44,367 | 7,910 | 388 | 5% |
| Vendor catalogues | 2,991,988 | 24,073 | 708 | 3% |
Synthetic Tractability Prediction
Many VEHICLe ring systems are clearly impractical (e.g., rings composed almost entirely of nitrogen). To separate plausible candidates from outlandish ones, the authors train a random forest classifier using the NovoD ArborPharm decision tree software (NovoDynamics, Inc.) within Pipeline Pilot:
- Features: ECFP_2 circular fingerprints (346 unique fragment types across VEHICLe), recording the presence or absence of each small substructure fragment per ring system
- Training labels: “Good” (769 ring systems found in compound databases totaling 3M+ molecules) vs. “bad” (24,098 remaining)
- Method: 100 trees using the Buja pure-bucket split method, optimized to minimize false negatives (GoodBias = 32, the ratio of bad to good examples). The PreserveMinority parameter was set to true, ensuring that training data selected for exclusion came exclusively from the “bad” class.
- Tree depth: 200 layers, chosen by systematic variation (50 to 250 in steps of 50) showing diminishing returns beyond this depth
- Node parameters: EnrichmentThreshold = 0.2 (if $\geq 20%$ of molecules in a node are “good”, the whole node is classified as good); minimum bucket size = 10 molecules per node ($0.04%$ of the dataset)
The classifier produces a $p(\text{good})$ score for each ring system. All 769 known ring systems scored $p(\text{good}) > 0.9$. Of the unknown ring systems, 2,185 (9%) were predicted tractable ($p(\text{good}) > 0.5$).
Validation: 36 VEHICLe rings from UCB’s corporate collection (not in the training set) were all correctly classified as good ($p(\text{good}) \geq 0.95$). Against the Beilstein database, 663 of 2,185 predicted-good unknowns had at least one substructure hit (30% minimum true positive rate), compared to only 374 of 21,913 predicted-bad unknowns (2% false negative rate), a 15-fold improvement over random. Selecting only $p(\text{good}) = 1.0$ predictions raised this ratio to 56-fold.
A final random forest incorporating Beilstein data predicted 3,288 unique unknown ring systems as tractable, with 232 having fewer than five heteroatoms and $p(\text{good}) > 0.95$. The authors manually selected 22 of these as “unconquered” challenges for synthetic chemists.
Ring System Usage Patterns
Analysis of ring system frequency across compound databases reveals striking concentration:
- Phenyl dominance: 2% of ring systems (15 types) account for 90% of occurrences, with phenyl alone at 70%.
- Heteroatom penalty: The significance of ring system usage drops sharply with increasing heteroatom count, quantified as:
$$ \text{significance}_{i,j} = \frac{\text{nobs}_{i,j} / \text{nobs}_{j}}{\text{ntot}_{i,j} / \text{ntot}_{j}} $$
where $i$ is the number of heteroatoms, $j$ is the compound set, $\text{nobs}$ is the frequency of observation, and $\text{ntot}$ is the total count in VEHICLe. Drug molecules in clinical trials show an even steeper drop-off than the broader compound set.
- Frequency distribution: Ring system frequency does not follow Zipf’s power law across the full range. Only ring systems occurring fewer than 500 times follow a power-law distribution.
- Publication rate decline: The rate of first publication of novel heteroaromatic ring systems peaked at about 41 per year in the late 1970s and declined to 5-10 per year by the early 2000s.
The concentration likely reflects the “principle of least effort,” the phylogenetic nature of drug discovery, and conservative risk management in pharma, rather than inherent unsuitability of the unused ring systems.
Reproducibility Details
The enumeration method is fully described and could be reimplemented, but the original implementation relies on proprietary software. The random forest model also uses proprietary tools but is specified in sufficient detail for reproduction with open-source alternatives.
| Artifact | Type | License | Notes |
|---|---|---|---|
| VEHICLe on Wolfram Data Repository | Dataset | Unknown | 24,867 ring systems with 16 properties each |
- Software dependencies: Pipeline Pilot (Accelrys Software Inc.) for enumeration; NovoD ArborPharm (NovoDynamics, Inc.) for decision trees. Both are proprietary.
- Hardware: 3 GHz Intel Xeon workstation (enumeration completes in ~3 minutes).
- Missing components: Original Pipeline Pilot protocols and rxn files are not publicly released. ECFP_2 fingerprints used a proprietary Accelrys implementation, though open-source equivalents (RDKit Morgan fingerprints with radius 1) exist.
- Reproducibility status: Partially Reproducible. The VEHICLe library itself is publicly available, and the method is described in sufficient detail for reimplementation with modern open-source tools, but the original code and protocols are not released.
Paper Information
- Journal: Journal of Medicinal Chemistry, Vol. 52, No. 9, pp. 2952-2963
- Published: April 6, 2009
@article{pitt2009heteroaromatic,
title={Heteroaromatic Rings of the Future},
author={Pitt, William R. and Parry, David M. and Perry, Benjamin G. and Groom, Colin R.},
journal={Journal of Medicinal Chemistry},
volume={52},
number={9},
pages={2952--2963},
year={2009},
publisher={American Chemical Society},
doi={10.1021/jm801513z}
}